Modeling the adenosine system as a modulator of cognitive performance and sleep patterns during sleep restriction and recovery
- PMID: 29073206
- PMCID: PMC5675465
- DOI: 10.1371/journal.pcbi.1005759
Modeling the adenosine system as a modulator of cognitive performance and sleep patterns during sleep restriction and recovery
Abstract
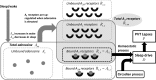
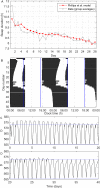
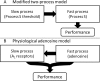
Sleep loss causes profound cognitive impairments and increases the concentrations of adenosine and adenosine A1 receptors in specific regions of the brain. Time courses for performance impairment and recovery differ between acute and chronic sleep loss, but the physiological basis for these time courses is unknown. Adenosine has been implicated in pathways that generate sleepiness and cognitive impairments, but existing mathematical models of sleep and cognitive performance do not explicitly include adenosine. Here, we developed a novel receptor-ligand model of the adenosine system to test the hypothesis that changes in both adenosine and A1 receptor concentrations can capture changes in cognitive performance during acute sleep deprivation (one prolonged wake episode), chronic sleep restriction (multiple nights with insufficient sleep), and subsequent recovery. Parameter values were estimated using biochemical data and reaction time performance on the psychomotor vigilance test (PVT). The model closely fit group-average PVT data during acute sleep deprivation, chronic sleep restriction, and recovery. We tested the model's ability to reproduce timing and duration of sleep in a separate experiment where individuals were permitted to sleep for up to 14 hours per day for 28 days. The model accurately reproduced these data, and also correctly predicted the possible emergence of a split sleep pattern (two distinct sleep episodes) under these experimental conditions. Our findings provide a physiologically plausible explanation for observed changes in cognitive performance and sleep during sleep loss and recovery, as well as a new approach for predicting sleep and cognitive performance under planned schedules.
Conflict of interest statement
EBK received travel reimbursement from the Sleep Technology Council and has served as an expert witness in cases involving transportation safety and sleep deprivation.
Figures


References
-
- Jewett ME, Dijk DJ, Kronauer RE, Dinges DF. Dose-response relationship between sleep duration and human psychomotor vigilance and subjective alertness. Sleep. 1999; 22: 171–179. - PubMed
-
- Drummond SPA, Paulus MP, Tapert SF. Effects of two nights sleep deprivation and two nights recovery sleep on response inhibition. J Sleep Res. 2006; 15: 261–265. doi: 10.1111/j.1365-2869.2006.00535.x - DOI - PubMed
-
- Van Dongen HPA, Maislin G, Mullington JM, Dinges DF. The cumulative cost of additional wakefulness: Dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep. 2003; 26: 117–126. - PubMed
-
- Belenky G, Wesenstein NJ, Thorne DR, Thomas ML, Sing HC, Redmond DP, et al. Patterns of performance degradation and restoration during sleep restriction and subsequent recovery: A sleep dose-response study. J Sleep Res. 2003; 12: 1–12. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

