Luciferase Activity of Insect Fatty Acyl-CoA Synthetases with Synthetic Luciferins
- PMID: 29073357
- PMCID: PMC5836727
- DOI: 10.1021/acschembio.7b00813
Luciferase Activity of Insect Fatty Acyl-CoA Synthetases with Synthetic Luciferins
Abstract
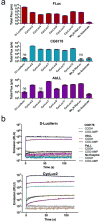
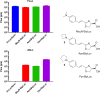
Long-chain fatty acyl-CoA synthetases (ACSLs) are homologues of firefly luciferase but are incapable of emitting light with firefly luciferin. Recently, we found that an ACSL from the fruit fly Drosophila melanogaster is a latent luciferase that will emit light with the synthetic luciferin CycLuc2. Here, we have profiled a panel of three insect ACSLs with a palette of >20 luciferin analogues. An ACSL from the nonluminescent beetle Agrypnus binodulus (AbLL) was found to be a second latent luciferase with distinct substrate specificity. Several rigid luciferins emit light with both ACSLs, but styryl luciferin analogues are light-emitting substrates only for AbLL. On the other hand, an ACSL from the luminescent beetle Pyrophorus angustus lacks luciferase activity with all tested analogues, despite its higher homology to beetle luciferases. Further study of ACSLs is expected to shed light on the features necessary for bioluminescence and substrate selectivity.
Figures

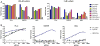
Similar articles
-
Lessons Learned from Luminous Luciferins and Latent Luciferases.ACS Chem Biol. 2018 Jul 20;13(7):1734-1740. doi: 10.1021/acschembio.7b00964. Epub 2018 Feb 19. ACS Chem Biol. 2018. PMID: 29439568 Free PMC article. Review.
-
Orthologous gene of beetle luciferase in non-luminous click beetle, Agrypnus binodulus (Elateridae), encodes a fatty acyl-CoA synthetase.Gene. 2008 Jan 15;407(1-2):169-75. doi: 10.1016/j.gene.2007.10.013. Epub 2007 Oct 13. Gene. 2008. PMID: 17996401
-
Evaluating Brightness and Spectral Properties of Click Beetle and Firefly Luciferases Using Luciferin Analogues: Identification of Preferred Pairings of Luciferase and Substrate for In Vivo Bioluminescence Imaging.Mol Imaging Biol. 2020 Dec;22(6):1523-1531. doi: 10.1007/s11307-020-01523-7. Epub 2020 Sep 14. Mol Imaging Biol. 2020. PMID: 32926287 Free PMC article.
-
Brominated Luciferins Are Versatile Bioluminescent Probes.Chembiochem. 2017 Jan 3;18(1):96-100. doi: 10.1002/cbic.201600564. Epub 2016 Dec 8. Chembiochem. 2017. PMID: 27930848 Free PMC article.
-
Firefly luciferase: an adenylate-forming enzyme for multicatalytic functions.Cell Mol Life Sci. 2010 Feb;67(3):387-404. doi: 10.1007/s00018-009-0170-8. Epub 2009 Oct 27. Cell Mol Life Sci. 2010. PMID: 19859663 Free PMC article. Review.
Cited by
-
Sulfonamides Are an Overlooked Class of Electron Donors in Luminogenic Luciferins and Fluorescent Dyes.Org Lett. 2019 Mar 15;21(6):1641-1644. doi: 10.1021/acs.orglett.9b00173. Epub 2019 Mar 5. Org Lett. 2019. PMID: 30835125 Free PMC article.
-
A Comprehensive Exploration of Bioluminescence Systems, Mechanisms, and Advanced Assays for Versatile Applications.Biochem Res Int. 2024 Feb 5;2024:8273237. doi: 10.1155/2024/8273237. eCollection 2024. Biochem Res Int. 2024. PMID: 38347947 Free PMC article. Review.
-
Lessons Learned from Luminous Luciferins and Latent Luciferases.ACS Chem Biol. 2018 Jul 20;13(7):1734-1740. doi: 10.1021/acschembio.7b00964. Epub 2018 Feb 19. ACS Chem Biol. 2018. PMID: 29439568 Free PMC article. Review.
-
Caged luciferins for bioluminescent activity-based sensing.Curr Opin Biotechnol. 2019 Dec;60:198-204. doi: 10.1016/j.copbio.2019.05.002. Epub 2019 Jun 11. Curr Opin Biotechnol. 2019. PMID: 31200275 Free PMC article. Review.
-
Enzymatic promiscuity and the evolution of bioluminescence.FEBS J. 2020 Apr;287(7):1369-1380. doi: 10.1111/febs.15176. Epub 2019 Dec 27. FEBS J. 2020. PMID: 31828943 Free PMC article. Review.
References
-
- Takakura H, Kojima R, Kamiya M, Kobayashi E, Komatsu T, Ueno T, Terai T, Hanaoka K, Nagano T, Urano Y. New class of bioluminogenic probe based on bioluminescent enzyme-induced electron transfer: BioLeT. J Am Chem Soc. 2015;137:4010–4013. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

