Ferritin is secreted via 2 distinct nonclassical vesicular pathways
- PMID: 29074498
- PMCID: PMC5774206
- DOI: 10.1182/blood-2017-02-768580
Ferritin is secreted via 2 distinct nonclassical vesicular pathways
Abstract
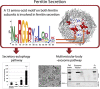
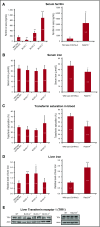
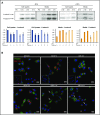
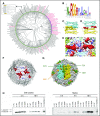
Ferritin turnover plays a major role in tissue iron homeostasis, and ferritin malfunction is associated with impaired iron homeostasis and neurodegenerative diseases. In most eukaryotes, ferritin is considered an intracellular protein that stores iron in a nontoxic and bioavailable form. In insects, ferritin is a classically secreted protein and plays a major role in systemic iron distribution. Mammalian ferritin lacks the signal peptide for classical endoplasmic reticulum-Golgi secretion but is found in serum and is secreted via a nonclassical lysosomal secretion pathway. This study applied bioinformatics and biochemical tools, alongside a protein trafficking mouse models, to characterize the mechanisms of ferritin secretion. Ferritin trafficking via the classical secretion pathway was ruled out, and a 2:1 distribution of intracellular ferritin between membrane-bound compartments and the cytosol was observed, suggesting a role for ferritin in the vesicular compartments of the cell. Focusing on nonclassical secretion, we analyzed mouse models of impaired endolysosomal trafficking and found that ferritin secretion was decreased by a BLOC-1 mutation but increased by BLOC-2, BLOC-3, and Rab27A mutations of the cellular trafficking machinery, suggesting multiple export routes. A 13-amino-acid motif unique to ferritins that lack the secretion signal peptide was identified on the BC-loop of both subunits and plays a role in the regulation of ferritin secretion. Finally, we provide evidence that secretion of iron-rich ferritin was mediated via the multivesicular body-exosome pathway. These results enhance our understanding of the mechanism of ferritin secretion, which is an important piece in the puzzle of tissue iron homeostasis.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures



Similar articles
-
Trafficking of a multifunctional protein by endosomal microautophagy: linking two independent unconventional secretory pathways.FASEB J. 2019 Apr;33(4):5626-5640. doi: 10.1096/fj.201802102R. Epub 2019 Jan 14. FASEB J. 2019. PMID: 30640524
-
Serum ferritin is derived primarily from macrophages through a nonclassical secretory pathway.Blood. 2010 Sep 2;116(9):1574-84. doi: 10.1182/blood-2009-11-253815. Epub 2010 May 14. Blood. 2010. PMID: 20472835
-
Decoupling ferritin synthesis from free cytosolic iron results in ferritin secretion.Cell Metab. 2011 Jan 5;13(1):57-67. doi: 10.1016/j.cmet.2010.12.003. Cell Metab. 2011. Retraction in: Cell Metab. 2012 Jun 6;15(6):927. doi: 10.1016/j.cmet.2012.04.012. PMID: 21195349 Free PMC article. Retracted.
-
A possible role for secreted ferritin in tissue iron distribution.J Neural Transm (Vienna). 2011 Mar;118(3):337-47. doi: 10.1007/s00702-011-0582-0. Epub 2011 Feb 6. J Neural Transm (Vienna). 2011. PMID: 21298454 Review.
-
New iron export pathways acting via holo-ferritin secretion.Arch Biochem Biophys. 2023 Sep 15;746:109737. doi: 10.1016/j.abb.2023.109737. Epub 2023 Sep 7. Arch Biochem Biophys. 2023. PMID: 37683905 Review.
Cited by
-
Ferritinophagy: A novel insight into the double-edged sword in ferritinophagy-ferroptosis axis and human diseases.Cell Prolif. 2024 Jul;57(7):e13621. doi: 10.1111/cpr.13621. Epub 2024 Feb 23. Cell Prolif. 2024. PMID: 38389491 Free PMC article. Review.
-
Increased Gene Expression of RUNX2 and SOX9 in Mesenchymal Circulating Progenitors Is Associated with Autophagy during Physical Activity.Oxid Med Cell Longev. 2019 Oct 15;2019:8426259. doi: 10.1155/2019/8426259. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31737174 Free PMC article.
-
Co-regulation of hepatic steatosis by ferritinophagy and unsaturated fatty acid supply.Hepatol Commun. 2022 Oct;6(10):2640-2653. doi: 10.1002/hep4.2040. Epub 2022 Jul 21. Hepatol Commun. 2022. PMID: 35861547 Free PMC article.
-
Extracellular vesicles in type 2 diabetes mellitus: key roles in pathogenesis, complications, and therapy.J Extracell Vesicles. 2019 Jun 14;8(1):1625677. doi: 10.1080/20013078.2019.1625677. eCollection 2019. J Extracell Vesicles. 2019. PMID: 31258879 Free PMC article. Review.
-
Iron-Deficiency in Atopic Diseases: Innate Immune Priming by Allergens and Siderophores.Front Allergy. 2022 May 10;3:859922. doi: 10.3389/falgy.2022.859922. eCollection 2022. Front Allergy. 2022. PMID: 35769558 Free PMC article. Review.
References
-
- Anderson GJ. Ironing out disease: inherited disorders of iron homeostasis. IUBMB Life. 2001;51(1):11-17. - PubMed
-
- Frazer DM, Anderson GJ. The regulation of iron transport. Biofactors. 2014;40(2):206-214. - PubMed
-
- Knutson M, Wessling-Resnick M. Iron metabolism in the reticuloendothelial system. Crit Rev Biochem Mol Biol. 2003;38(1):61-88. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

