Regulation of reproduction and longevity by nutrient-sensing pathways
- PMID: 29074705
- PMCID: PMC5748989
- DOI: 10.1083/jcb.201707168
Regulation of reproduction and longevity by nutrient-sensing pathways
Abstract
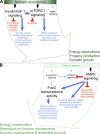
Nutrients are necessary for life, as they are a crucial requirement for biological processes including reproduction, somatic growth, and tissue maintenance. Therefore, signaling systems involved in detecting and interpreting nutrient or energy levels-most notably, the insulin/insulin-like growth factor 1 (IGF-1) signaling pathway, mechanistic target of rapamycin (mTOR), and adenosine monophosphate-activated protein kinase (AMPK)-play important roles in regulating physiological decisions to reproduce, grow, and age. In this review, we discuss the connections between reproductive senescence and somatic aging and give an overview of the involvement of nutrient-sensing pathways in controlling both reproductive function and lifespan. Although the molecular mechanisms that affect these processes can be influenced by distinct tissue-, temporal-, and pathway-specific signaling events, the progression of reproductive aging and somatic aging is systemically coordinated by integrated nutrient-sensing signaling pathways regulating somatic tissue maintenance in conjunction with reproductive capacity.
© 2018 Templeman and Murphy.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

