Metformin for endometrial hyperplasia
- PMID: 29077194
- PMCID: PMC6485333
- DOI: 10.1002/14651858.CD012214.pub2
Metformin for endometrial hyperplasia
Update in
-
Metformin for endometrial hyperplasia.Cochrane Database Syst Rev. 2024 May 2;5(5):CD012214. doi: 10.1002/14651858.CD012214.pub3. Cochrane Database Syst Rev. 2024. PMID: 38695827 Free PMC article.
Abstract
Background: Endometrial cancer is one of the most common gynaecological cancers in the world. Rates of endometrial cancer are rising, in part because of rising obesity rates. Endometrial hyperplasia is a precancerous condition in women that can lead to endometrial cancer if left untreated. Endometrial hyperplasia occurs more commonly than endometrial cancer. Progesterone tablets currently used to treat women with endometrial hyperplasia are associated with adverse effects in up to 84% of women. The levonorgestrel intrauterine device (Mirena Coil, Bayer HealthCare Pharmaceuticals, Inc., Whippany, NJ, USA) may improve compliance, but it is invasive, is not acceptable to all women, and is associated with irregular vaginal bleeding in 82% of cases. Therefore, an alternative treatment for women with endometrial hyperplasia is needed. Metformin, a drug that is often used to treat people with diabetes, has been shown in some human studies to reverse endometrial hyperplasia. However, the effectiveness and safety of metformin for treatment of endometrial hyperplasia remain uncertain.
Objectives: To determine the effectiveness and safety of metformin in treating women with endometrial hyperplasia.
Search methods: We searched the Cochrane Gynaecology and Fertility Specialised Register, the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, the Cumulative Index to Nursing and Allied Health Literature (CINAHL), PubMed, Google Scholar, OpenGrey, Latin American Caribbean Health Sciences Literature (LILACS), and two trials registers from inception to 10 January 2017. We searched the bibliographies of all included studies and reviews on this topic. We also handsearched the conference abstracts of the European Society of Human Reproduction and Embryology (ESHRE) 2015 and the American Society for Reproductive Medicine (ASRM) 2015.
Selection criteria: We included randomised controlled trials (RCTs) and cross-over trials comparing metformin (used alone or in combination with other medical therapies) versus placebo or no treatment, any conventional medical treatment, or any other active intervention for women with histologically confirmed endometrial hyperplasia of any type.
Data collection and analysis: Two review authors independently assessed studies for eligibility, extracted data from included studies, and assessed the risk of bias of included studies. We resolved disagreements by discussion or by deferment to a third review author. When study details were missing, review authors contacted study authors. The primary outcome of this review was regression of endometrial hyperplasia histology (with or without atypia) towards normal histology. Secondary outcome measures included recurrence of endometrial hyperplasia, progression of endometrial hyperplasia to endometrial cancer, hysterectomy rate, abnormal uterine bleeding, health-related quality of life, and adverse effects during treatment.
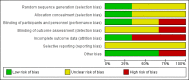
Main results: We included three RCTs in which a total of 77 women took part. We rated the quality of the evidence as very low for all outcomes owing to very serious risk of bias (associated with poor reporting, attrition, and limitations in study design) and imprecision.We performed a meta-analysis of two trials with 59 participants. When metformin was compared with megestrol acetate in women with endometrial hyperplasia, we found insufficient evidence to determine whether there were differences between groups for the following outcomes: regression of endometrial hyperplasia histology towards normal histology (odds ratio (OR) 3.34, 95% confidence interval (CI) 0.97 to 11.57, two RCTs, n = 59, very low-quality evidence), hysterectomy rates (OR 0.91, 95% CI 0.05 to 15.52, two RCTs, n = 59, very low-quality evidence), and rates of abnormal uterine bleeding (OR 0.91, 95% CI 0.05 to 15.52, two RCTs, n = 44 , very low-quality evidence). We found no data for recurrence of endometrial hyperplasia or health-related quality of life. Both studies (n = 59) provided data on progression of endometrial hyperplasia to endometrial cancer as well as one (n = 16) reporting some adverse effects in the metformin arm, notably nausea, thrombosis, lactic acidosis, abnormal liver and renal function among others.Another trial including 16 participants compared metformin plus megestrol acetate versus megestrol acetate alone in women with endometrial hyperplasia. We found insufficient evidence to determine whether there were differences between groups for the following outcomes: regression of endometrial hyperplasia histology towards normal histology (OR 9.00, 95% CI 0.94 to 86.52, one RCT, n = 16, very low-quality evidence), recurrence of endometrial hyperplasia among women who achieve regression (OR not estimable, no events recorded, one RCT, n = 8, very low-quality evidence), progression of endometrial hyperplasia to endometrial cancer (OR not estimable, no events recorded, one RCT, n = 13, very low-quality evidence), or hysterectomy rates (OR 0.29, 95% CI 0.01 to 8.37, one RCT, n = 16, very low-quality evidence). Investigators provided no data on abnormal uterine bleeding or health-related quality of life. In terms of adverse effects, three of eight participants (37.5%) in the metformin plus megestrol acetate study arm reported nausea.
Authors' conclusions: At present, evidence is insufficient to support or refute the use of metformin alone or in combination with standard therapy - specifically, megestrol acetate - versus megestrol acetate alone, for treatment of endometrial hyperplasia. Robustly designed and adequately powered randomised controlled trials yielding long-term outcome data are needed to address this clinical question.
Conflict of interest statement
WA is a Clinical Associate Professor and a Consultant Gynaecologist at Queen's Medical Centre, at Nottingham, in the UK. He previously submitted an application to the Nottingham Clinical Trials Unit (in the process of developing a research grant application to the UK NIHR) to conduct a clinical trial comparing metformin with progesterone for treatment of endometrial hyperplasia. A systematic review was suggested to support the trial.
Figures
References
References to studies included in this review
Shan 2014 {published and unpublished data}
-
- Shan W, Wang C, Zhang Z, Gu C, Ning C, Luo X, et al. Conservative therapy with metformin plus megestrol acetate for endometrial atypical hyperplasia: preliminary results of a pilot study. International Journal of Gynecological Cancer. Philadelphia: Lippincott Williams & Wilkins, 2014; Vol. 4:1306‐7.
Sharifzadeh 2016 {published data only}
Tabrizi 2014 {published and unpublished data}
-
- Tabrizi AD, Melli MS, Foroughi M, Ghojazadeh M, Bidadi S. Antiproliferative effect of metformin on the endometrium ‐ a clinical trial. Asian Pacific Journal of Cancer Prevention 2014;15(23):10067‐70. - PubMed
References to studies excluded from this review
Campagnoli 2013 {published data only}
-
- Campagnoli C, Abbà C, Ambroggio S, Brucato T, Pasanisi P. Life‐style and metformin for the prevention of endometrial pathology in postmenopausal women. Gynecological Endocrinology 2013;29(2):119‐24. - PubMed
IRCT201412085563N6, 2015 {published data only}
-
- IRCT201412085563N6, Vice Chancellor for Research Tabriz University of Medical Sciences. The effect of letrozole and metformin on endometrial histology in patients with disordered proliferative endometrium and simple hyperplasia uterus cancer. www.irct.ir 2015.
Legro 2007 {published data only}
-
- Legro RS, Zaino RJ, Demers LM, Kunselman AR, Gnatuk CL, Williams NI, et al. The effects of metformin and rosiglitazone, alone and in combination, on the ovary and endometrium in polycystic ovary syndrome. American Journal of Obstetrics and Gynecology 2007;196(4):402.e1‐402.e11. - PubMed
Mitsuhashi 2014 {published data only}
-
- Mitsuhashi A, Kiyokawa T, Sato Y, Shozu M. Effects of metformin on endometrial cancer cell growth in vivo: a preoperative prospective trial. Cancer 2014;120(19):2986‐95. - PubMed
Mitsuhashi 2016 {published data only}
-
- Mitsuhashi A, Sato Y, Kiyokawa T, Koshizaka M, Hanaoka H, Shozu M. Phase II study of medroxyprogesterone acetate plus metformin as a fertility‐sparing treatment for atypical endometrial hyperplasia and endometrial cancer. Annals of Oncology 2016;27(2):262‐6. - PubMed
NCT01685762 {published and unpublished data}
-
- NCT01685762. UNC Lineberger Comprehensive Cancer Center. Metformin for the Treatment of Endometrial Hyperplasia. clinicaltrials.gov 2017.
NCT02035787, 2014 {published and unpublished data}
-
- NCT02035787. UNC Lineberger Comprehensive Cancer Center. Metformin With the Levonorgestrel‐Releasing Intrauterine Device for the Treatment of Complex Atypical Hyperplasia (CAH) and Endometrial Cancer (EC) in Non‐surgical Patients. clinicaltrials.gov 2014.
Perez‐Lopez 2014 {published data only}
-
- Perez‐Lopez FR. Metformin treatment and evolution of endometrial cancer. Climacteric 2014;17(2):207‐9. - PubMed
Randall 2014 {published data only}
-
- Randall LM, Gibson SJ, Monk BJ. The American Society of Clinical Oncology 50th Annual Meeting 2014: an overview and summary of selected abstracts. Gynecologic Oncology 2014;134(2):222‐7. - PubMed
Shen 2008 {published data only}
-
- Shen ZQ, Zhu HT, Lin JF. Reverse of progestin‐resistant atypical endometrial hyperplasia by metformin and oral contraceptives. Obstetrics & Gynecology 2008;112(2):465‐7. - PubMed
Sivalingam 2015 {published data only}
-
- Sivalingam V, McVey R, Gilmour K, Ali S, Roberts C, Renehan A, et al. A presurgical window‐of‐opportunity study of metformin in obesity‐driven endometrial cancer. The Lancet 2015;385 North American Edition:S90. - PubMed
Sivalingam 2016 {published data only}
Sivalingham 2015b {published data only}
-
- Sivalingama VN, McVeyb R, Kitsona S, Gilmourc K, Alid S, Robertsa C, et al. Pre‐surgical window study of metformin in obesity‐driven endometrial cancer. Gynecologic Oncology. 2015; Vol. 137(Suppl 1):18. [DOI: ]
Zhou 2015 {published data only}
-
- Zhou R, Yang Y, Lu Q, Wang J, Miao Y, Wang S, et al. Prognostic factors of oncological and reproductive outcomes in fertility‐sparing treatment of complex atypical hyperplasia and low‐grade endometrial cancer using oral progestin in Chinese patients. Gynecologic Oncology 2015;139(3):424‐8. - PubMed
References to ongoing studies
IRCT201410275283N11 {published data only (unpublished sought but not used)}
-
- IRCT201410275283N11. Comparison of metformin‐megestrol combination and megestrol alone on endometrial histology in patients with disordered proliferative or hyperplastic endometrium.. http://en.search.irct.ir/view/20539 (first received 28 November 2014).
NCT01686126 {published data only (unpublished sought but not used)}
-
- NCT01686126. Improving the treatment for women with early stage cancer of the uterus (feMMe). https://clinicaltrials.gov/ct2/show/NCT01686126 (first received 12 September 2012).
NCT01968317 {published data only (unpublished sought but not used)}
-
- NCT01968317. Megestrol acetate plus metformin to megestrol acetate in patients with endometrial atypical hyperplasia or early stage endometrial adenocarcinoma. https://clinicaltrials.gov/ct2/show/NCT01968317 (first received 16 October 2013).
Additional references
Alimova 2009
-
- Alimova IN, Liu B, Fan Z, Edgerton SM, Dillon T, Lind SE, et al. Metformin inhibits breast cancer cell growth, colony formation and induces cell cycle arrest in vitro. Cell Cycle (Georgetown, Tex) 2009;8(6):909‐15. [PUBMED: 19221498] - PubMed
Ben 2011
-
- Ben Sahra I, Regazzetti C, Robert G, Laurent K, Marchand‐Brustel Y, Auberger P, et al. Metformin, independent of AMPK, induces mTOR inhibition and cell‐cycle arrest through REDD1. Cancer Research 2011;71(13):4366‐72. [PUBMED: 21540236] - PubMed
BNF 2017
-
- Joint Formulary Committee. British National Formulary. 3rd Edition. London: BMJ Group and Pharmaceutical Press, 2017.
Chang 2002
-
- Chang RJ, Heindel JJ, Dunaif A. Polcystic ovarian syndrome. Polycystic Ovarian Syndrome. New York: Marcel Dekker Inc., 2002.
Chlebowski 2012
Cohen 1988
-
- Cohen J. Statistical Power Analysis for the Behavioural Sciences. 2nd Edition. Hillsdale, NJ: Lawrence Eribaum, 1988.
Costello 2007
-
- Costello M, Shrestha B, Eden J, Sjoblom P, Johnson N. Insulin‐sensitising drugs versus the combined oral contraceptive pill for hirsutism, acne and risk of diabetes, cardiovascular disease, and endometrial cancer in polycystic ovary syndrome. The Cochrane Database of Systematic Reviews 2007;1(1):CD005552. [PUBMED: 17253562] - PubMed
Covidence [Computer program]
-
- Veritas Health Innovation. Covidence. Version accessed 10 January 2017. Melbourne: Veritas Health Innovation.
de Jager 2010
Dowling 2011
Erdemoglu 2009
-
- Erdemoglu E, Güney M, Giray SG, Take G, Mungan T. Effects of metformin on mammalian target of rapamycin in a mouse model of endometrial hyperplasia. European Journal of Obstetrics & Gynecology and Reproductive Biology 2009;145(2):195‐9. - PubMed
ESHRE/ASRM 2004
-
- Rotterdam ESHRE/ASRM‐Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long‐term health risks related to polycystic ovary syndrome. Fertility and Sterility 2004;81(1):19‐25. [PUBMED: 14711538] - PubMed
Gallos 2013
-
- Gallos ID, Krishan P, Shehmar M, Ganesan R, Gupta JK. Relapse of endometrial hyperplasia after conservative treatment: a cohort study with long‐term follow‐up. Human Reproduction (Oxford, England) 2013;28(5):1231‐6. [PUBMED: 23466671] - PubMed
GRADEpro GDT 2015 [Computer program]
-
- McMaster University (developed by Evidence Prime). GRADEproGDT. Version accessed 10 May 2017. Hamilton (ON): McMaster University (developed by Evidence Prime), 2015.
Haoula 2012
-
- Haoula Z, Salman M, Atiomo W. Evaluating the association between endometrial cancer and polycystic ovary syndrome. Human Reproduction (Oxford, England) 2012;27(5):1327‐31. [PUBMED: 22367984] - PubMed
Higgins 2003
Higgins 2011
-
- Higgins JP, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Holm 2012
-
- Holm NS, Glintborg D, Andersen MS, Schledermann D, Ravn P. The prevalence of endometrial hyperplasia and endometrial cancer in women with polycystic ovary syndrome or hyperandrogenism. Acta Obstetricia et Gynecologica Scandinavica 2012;91(10):1173‐6. [PUBMED: 22583042] - PubMed
Kurman 1985
-
- Kurman RJ, Kaminski PF, Norris HJ. The behavior of endometrial hyperplasia. A long‐term study of "untreated" hyperplasia in 170 patients. Cancer 1985;56(2):403‐12. [PUBMED: 4005805] - PubMed
Lacey 2010
Moher 2009
Nevadunsky 2014
RevMan 2014 [Computer program]
-
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Rosato 2011
-
- Rosato V, Zucchetto A, Bosetti C, Dal Maso L, Montella M, Pelucchi C, et al. Metabolic syndrome and endometrial cancer risk. Annals of Oncology 2011;22(4):884‐9. [PUBMED: 20937645] - PubMed
Rudnicka 2009
-
- Rudnicka E, Wierzba W, Radowicki S. Evaluation of endometrial histologic morphology in patients with polycystic ovary syndrome [Ocena obrazow morfologicznych endometrium u pacjentek z zespolem policystycznych jajnikow.]. Ginekologia Polska 2009;80(2):103‐6. [PUBMED: 19338206] - PubMed
Session 2003
Shafiee 2014
-
- Shafiee M, Khan G, Ariffin R, Abu J, Chapman C, Deen S, et al. Preventing endometrial caner risk in polycystic ovarian syndrome (PCOS) women: could metformin help?. Gynecologic Oncology 2014;132:248‐53. - PubMed
Torres 2012
Vrachnis 2016
-
- Vrachnis N, Lavazzo C, Iliodromiti Z, Sifakis S, Alexandrou A, Siristatidis C, et al. Diabetes mellitus and gynecologic cancer: molecular mechanisms, epidemiological, clinical and prognostic perspectives. Archives of Gynaecology and Obstetrics 2016;293(2):239‐46. - PubMed
Yang 2011
Zaino 2014
-
- Zaino R, Carinelli SG, Ellenson LH. Tumours of the Uterine Corpus: Epithelial Tumours and Precursors. In: Kurman RJ, Carcangiu ML, Herrington CS, Young RH editor(s). WHO Classification of Tumours of Female Reproductive Organs. 4th Edition. Vol. 6, Lyon: WHO Press, 2014:125‐6.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources