Host Cell Vimentin Restrains Toxoplasma gondii Invasion and Phosphorylation of Vimentin is Partially Regulated by Interaction with Tg ROP18
- PMID: 29104504
- PMCID: PMC5666328
- DOI: 10.7150/ijbs.21247
Host Cell Vimentin Restrains Toxoplasma gondii Invasion and Phosphorylation of Vimentin is Partially Regulated by Interaction with Tg ROP18
Abstract
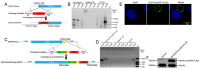
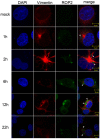
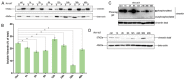
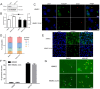
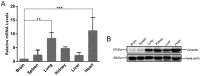
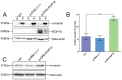
The obligate intracellular parasite, Toxoplasma gondii, manipulates the cytoskeleton of its host cells to facilitate infection. A significant rearrangement of host cell vimentin around Toxoplasma parasitophorous vacuoles is observed during the course of infection. ROP18 (TgROP18) is a serine-threonine kinase secreted by T. gondii rhoptry and a major virulence factor; however, the mechanisms by which this kinase modulates host factors remain poorly understood. Different and dynamic patterns of vimentin solubility, phosphorylation, and expression levels were observed in host cells infected with T. gondii strain RH and RH Δrop18 strains, suggesting that TgROP18 contributes to the regulation of these dynamic patterns. Additionally, host cell vimentin was demonstrated to interact with and be phosphorylated by TgROP18. A significant increase in T. gondii infection rate was observed in vimentin knockout human brain microvessel endothelial cells (HBMEC), while vimentin knockout or knock down in host cells had no impact on parasite proliferation and egress. These results indicate that host cell vimentin can inhibit T. gondii invasion. Interestingly, western blotting of different mouse tissues indicated that the lowest vimentin expression level was present in the brain, which may explain the mechanism underlying the nervous system tropism of T. gondii, and the phenomenon of huge cyst burdens developing in the mouse brain during chronic infection.
Keywords: Phosphorylation; Reorganization; TgROP18; Toxoplasma gondii; Tropism.; Vimentin.
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures


Similar articles
-
TgROP18 targets IL20RB for host-defense-related-STAT3 activation during Toxoplasma gondii infection.Parasit Vectors. 2020 Aug 7;13(1):400. doi: 10.1186/s13071-020-04251-7. Parasit Vectors. 2020. PMID: 32767999 Free PMC article.
-
The Toxoplasma gondii Rhoptry Kinome Is Essential for Chronic Infection.mBio. 2016 May 10;7(3):e00193-16. doi: 10.1128/mBio.00193-16. mBio. 2016. PMID: 27165797 Free PMC article.
-
Toxoplasma gondii ROP18 inhibits human glioblastoma cell apoptosis through a mitochondrial pathway by targeting host cell P2X1.Parasit Vectors. 2019 Jun 4;12(1):284. doi: 10.1186/s13071-019-3529-1. Parasit Vectors. 2019. PMID: 31164145 Free PMC article.
-
[Research progress of interaction between Toxoplasma gondii rhoptry proteins and host cells].Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 2014 Aug;26(4):453-6. Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 2014. PMID: 25434152 Review. Chinese.
-
Research advances in interactions related to Toxoplasma gondii microneme proteins.Exp Parasitol. 2017 May;176:89-98. doi: 10.1016/j.exppara.2017.03.001. Epub 2017 Mar 9. Exp Parasitol. 2017. PMID: 28286325 Review.
Cited by
-
Genome-Wide Bimolecular Fluorescence Complementation-Based Proteomic Analysis of Toxoplasma gondii ROP18's Human Interactome Shows Its Key Role in Regulation of Cell Immunity and Apoptosis.Front Immunol. 2018 Feb 5;9:61. doi: 10.3389/fimmu.2018.00061. eCollection 2018. Front Immunol. 2018. PMID: 29459857 Free PMC article.
-
The molecular biophysics of extracellular vimentin and its role in pathogen-host interactions.Curr Opin Cell Biol. 2023 Dec;85:102233. doi: 10.1016/j.ceb.2023.102233. Epub 2023 Sep 5. Curr Opin Cell Biol. 2023. PMID: 37677998 Free PMC article. Review.
-
Toxoplasma gondii SAG1 targeting host cell S100A6 for parasite invasion and host immunity.iScience. 2021 Nov 26;24(12):103514. doi: 10.1016/j.isci.2021.103514. eCollection 2021 Dec 17. iScience. 2021. PMID: 34950858 Free PMC article.
-
[Effect of vimentin on activation of NLRP3 inflammasome in the brain of mice with EV71 infection].Nan Fang Yi Ke Da Xue Xue Bao. 2018 Jun 20;38(6):704-710. doi: 10.3969/j.issn.1673-4254.2018.06.10. Nan Fang Yi Ke Da Xue Xue Bao. 2018. PMID: 29997093 Free PMC article. Chinese.
-
Quantitative phosphoproteomic analysis of chicken DF-1 cells infected with Eimeria tenella, using tandem mass tag (TMT) and parallel reaction monitoring (PRM) mass spectrometry.Parasite. 2024;31:23. doi: 10.1051/parasite/2024027. Epub 2024 May 17. Parasite. 2024. PMID: 38759153 Free PMC article.
References
-
- Dubey JP. Toxoplasmosis of animals and humans. Florida, USA: CRC Press; 2010.
-
- Pappas G, Roussos N, Falagas ME. Toxoplasmosis snapshots: global status of Toxoplasma gondii seroprevalence and implications for pregnancy and congenital toxoplasmosis. Int J Parasitol. 2009;39:1385–94. - PubMed
-
- Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363:1965–76. - PubMed
-
- Elmore SA, Jones JL, Conrad PA. et al. Toxoplasma gondii: epidemiology, feline clinical aspects, and prevention. Trends Parasitol. 2010;26:190–6. - PubMed
-
- Morisaki JH, Heuser JE, Sibley LD. Invasion of Toxoplasma gondii occurs by active penetration of the host cell. J Cell Sci. 1995;108( Pt 6):2457–64. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

