Prodomain-growth factor swapping in the structure of pro-TGF-β1
- PMID: 29109152
- PMCID: PMC5798290
- DOI: 10.1074/jbc.M117.809657
Prodomain-growth factor swapping in the structure of pro-TGF-β1
Abstract
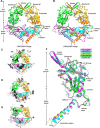
TGF-β is synthesized as a proprotein that dimerizes in the endoplasmic reticulum. After processing in the Golgi to cleave the N-terminal prodomain from the C-terminal growth factor (GF) domain in each monomer, pro-TGF-β is secreted and stored in latent complexes. It is unclear which prodomain and GF monomer are linked before proprotein convertase cleavage and how much conformational change occurs following cleavage. We have determined a structure of pro-TGF-β1 with the proprotein convertase cleavage site mutated to mimic the structure of the TGF-β1 proprotein. Structure, mutation, and model building demonstrate that the prodomain arm domain in one monomer is linked to the GF that interacts with the arm domain in the other monomer in the dimeric structure (i.e. the prodomain arm domain and GF domain in each monomer are swapped). Swapping has important implications for the mechanism of biosynthesis in the TGF-β family and is relevant to the mechanism for preferential formation of heterodimers over homodimers for some members of the TGF-β family. Our structure, together with two previous ones, also provides insights into which regions of the prodomain-GF complex are highly structurally conserved and which are perturbed by crystal lattice contacts.
Keywords: Arg-Gly-Asp-Leu-any-any-Leu/Ile (RGDLXX(L/I)); Protein Data Bank (PDB); activin; bone morphogenetic protein (BMP); crystal structure; dimerization; glycoprotein-A repetitions predominant protein (GARP); growth factor (GF); heterodimer; latency-associated peptide (LAP); latent TGF-β-binding proteins (LTBPs); prodomain; proprotein convertase; swapping; transforming growth factor beta (TGF-β).
© 2018 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures




References
-
- Gentry L. E., Webb N. R., Lim G. J., Brunner A. M., Ranchalis J. E., Twardzik D. R., Lioubin M. N., Marquardt H., and Purchio A. F. (1987) Type 1 transforming growth factor beta: amplified expression and secretion of mature and precursor polypeptides in Chinese hamster ovary cells. Mol. Cell Biol. 7, 3418–3427 10.1128/MCB.7.10.3418 - DOI - PMC - PubMed
-
- Wakefield L. M., Smith D. M., Flanders K. C., and Sporn M. B. (1988) Latent transforming growth factor-β from human platelets: a high molecular weight complex containing precursor sequences. J. Biol. Chem. 263, 7646–7654 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

