Aedes aegypti Molecular Responses to Zika Virus: Modulation of Infection by the Toll and Jak/Stat Immune Pathways and Virus Host Factors
- PMID: 29109710
- PMCID: PMC5660061
- DOI: 10.3389/fmicb.2017.02050
Aedes aegypti Molecular Responses to Zika Virus: Modulation of Infection by the Toll and Jak/Stat Immune Pathways and Virus Host Factors
Abstract
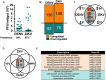
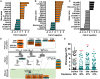
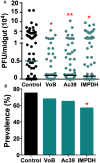
Zika (ZIKV) and dengue virus (DENV) are transmitted to humans by Aedes mosquitoes. However, the molecular interactions between the vector and ZIKV remain largely unexplored. In this work, we further investigated the tropism of ZIKV in two different Aedes aegypti strains and show that the virus infection kinetics, tissue migration, and susceptibility to infection differ between mosquito strains. We also compare the vector transcriptome changes upon ZIKV or DENV infection demonstrating that 40% of the mosquito's midgut infection-responsive transcriptome is virus-specific at 7 days after virus ingestion. Regulated genes included key factors of the mosquito's anti-viral immunity. Comparison of the ZIKV and DENV infection-responsive transcriptome data to those available for yellow fever virus and West Nile virus identified 26 genes likely to play key roles in virus infection of Aedes mosquitoes. Through reverse genetic analyses, we show that the Toll and the Jak/Stat innate immune pathways mediate increased resistance to ZIKV infection, and the conserved DENV host factors vATPase and inosine-5'-monophosphate dehydrogenase are also utilized for ZIKV infection.
Keywords: Aedes aegypti; Zika virus; dengue virus; innate immunity; vector competence.
Figures

References
-
- Adelman Z. N., Anderson M. A. E., Wiley M. R., Murreddu M. G., Samuel G. H., Morazzani E. M., et al. (2013). Cooler temperatures destabilize RNA interference and increase susceptibility of disease vector mosquitoes to viral infection. PLOS Negl. Trop. Dis. 7:e2239. 10.1371/journal.pntd.0002239 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

