Aprepitant Has Mixed Effects on Nausea and Reduces Other Symptoms in Patients With Gastroparesis and Related Disorders
- PMID: 29111115
- PMCID: PMC5742047
- DOI: 10.1053/j.gastro.2017.08.033
Aprepitant Has Mixed Effects on Nausea and Reduces Other Symptoms in Patients With Gastroparesis and Related Disorders
Abstract
Background & aims: There are few effective treatments for nausea and other symptoms in patients with gastroparesis and related syndromes. We performed a randomized trial of the ability of the neurokinin-1 receptor antagonist aprepitant to reduce symptoms in patients with chronic nausea and vomiting caused by gastroparesis or gastroparesis-like syndrome.
Methods: We conducted a 4-week multicenter, double-masked trial of 126 patients with at least moderate symptoms of chronic nausea and vomiting of presumed gastric origin for a minimum of 6 months. Patients were randomly assigned to groups given oral aprepitant (125 mg/day, n = 63) or placebo (n = 63). The primary outcome from the intention-to-treat analysis was reduction in nausea, defined as a decrease of 25 mm or more, or absolute level below 25 mm, on a daily patient-reported 0-to-100 visual analog scale (VAS) of nausea severity. We calculated relative risks of nausea improvement using stratified Cochran-Mental-Haenszel analysis.
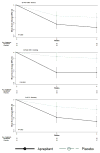
Results: Aprepitant did not reduce symptoms of nausea, based on the primary outcome measure (46% reduction in the VAS score in the aprepitant group vs 40% reduction in the placebo group; relative risk, 1.2; 95% CI, 0.8-1.7) (P = .43). However, patients in the aprepitant group had significant changes in secondary outcomes such as reduction in symptom severity (measured by the 0-5 Gastroparesis Clinical Symptom Index) for nausea (1.8 vs 1.0; P = .005), vomiting (1.6 vs 0.5; P = .001), and overall symptoms (1.3 vs 0.7; P = .001). Adverse events, predominantly mild or moderate in severity grade, were more common in aprepitant (22 of 63 patients, 35% vs 11 of 63, 17% in the placebo group) (P = .04).
Conclusions: In a randomized trial of patients with chronic nausea and vomiting caused by gastroparesis or gastroparesis-like syndrome, aprepitant did not reduce the severity of nausea when reduction in VAS score was used as the primary outcome. However, aprepitant had varying effects on secondary outcomes of symptom improvement. These findings support the need to identify appropriate patient outcomes for trials of therapies for gastroparesis, including potential additional trials for aprepitant. ClinicalTrials.gov no: NCT01149369.
Keywords: Aprepitant Treatment; Chronic Nausea and Vomiting; Gastroparesis; RCT.
Copyright © 2018 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
None of the authors report any conflict of interest with respect to this manuscript or the data in it
Figures
References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- U01 DK074008/DK/NIDDK NIH HHS/United States
- UL1 TR001105/TR/NCATS NIH HHS/United States
- U01 DK074007/DK/NIDDK NIH HHS/United States
- UL1 TR000093/TR/NCATS NIH HHS/United States
- U01 DK073985/DK/NIDDK NIH HHS/United States
- U01 DK073975/DK/NIDDK NIH HHS/United States
- U01 DK074035/DK/NIDDK NIH HHS/United States
- K23 DK103911/DK/NIDDK NIH HHS/United States
- UL1 TR000424/TR/NCATS NIH HHS/United States
- U01 DK073974/DK/NIDDK NIH HHS/United States
- UL1 TR000433/TR/NCATS NIH HHS/United States
- U24 DK074008/DK/NIDDK NIH HHS/United States
- U01 DK073983/DK/NIDDK NIH HHS/United States
- UL1 TR000135/TR/NCATS NIH HHS/United States
- U01 DK112193/DK/NIDDK NIH HHS/United States

