Consensus Treatment Plans for Chronic Nonbacterial Osteomyelitis Refractory to Nonsteroidal Antiinflammatory Drugs and/or With Active Spinal Lesions
- PMID: 29112802
- PMCID: PMC5938153
- DOI: 10.1002/acr.23462
Consensus Treatment Plans for Chronic Nonbacterial Osteomyelitis Refractory to Nonsteroidal Antiinflammatory Drugs and/or With Active Spinal Lesions
Abstract
Objective: To develop standardized treatment regimens for chronic nonbacterial osteomyelitis (CNO), also known as chronic recurrent multifocal osteomyelitis (CRMO), to enable comparative effectiveness treatment studies.
Methods: Virtual and face-to-face discussions and meetings were held within the CNO/CRMO subgroup of the Childhood Arthritis and Rheumatology Research Alliance (CARRA). A literature search was conducted, and CARRA membership was surveyed to evaluate available treatment data and identify current treatment practices. Nominal group technique was used to achieve consensus on treatment plans for CNO refractory to nonsteroidal antiinflammatory drug (NSAID) monotherapy and/or with active spinal lesions.
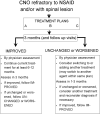
Results: Three consensus treatment plans (CTPs) were developed for the first 12 months of therapy for CNO patients refractory to NSAID monotherapy and/or with active spinal lesions. The 3 CTPs are methotrexate or sulfasalazine, tumor necrosis factor inhibitors with optional methotrexate, and bisphosphonates. Short courses of glucocorticoids and continuation of NSAIDs are permitted for all regimens. Consensus was achieved on these CTPs among CARRA members. Consensus was also reached on subject eligibility criteria, initial evaluations that should be conducted prior to the initiation of CTPs, and data items to collect to assess treatment response.
Conclusion: Three consensus treatment plans were developed for pediatric patients with CNO refractory to NSAIDs and/or with active spinal lesions. Use of these CTPs will provide additional information on efficacy and will generate meaningful data for comparative effectiveness research in CNO.
© 2017, American College of Rheumatology.
References
-
- Hofmann SR, Schnabel A, Rosen-Wolff A, Morbach H, Girschick HJ, Hedrich CM. Chronic Nonbacterial Osteomyelitis: Pathophysiological Concepts and Current Treatment Strategies. J Rheumatol. 2016 - PubMed
-
- Ringold S, Weiss PF, Colbert RA, DeWitt EM, Lee T, Onel K, Prahalad S, Schneider R, Shenoi S, Vehe RK KYJIARC of the CA and RRA Childhood Arthritis and Rheumatology Research Alliance Consensus Treatment Plans for New Onset Polyarticular Juvenile Idiopathic Arthritis. Arthritis Care Res (Hoboken) 2014;66:1063–72. doi: 10.1002/acr.22259. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical