Aberrant Rac1-cofilin signaling mediates defects in dendritic spines, synaptic function, and sensory perception in fragile X syndrome
- PMID: 29114038
- PMCID: PMC5988355
- DOI: 10.1126/scisignal.aan0852
Aberrant Rac1-cofilin signaling mediates defects in dendritic spines, synaptic function, and sensory perception in fragile X syndrome
Abstract
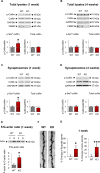
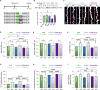
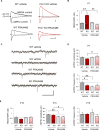
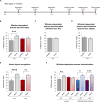
Fragile X syndrome (FXS) is the most common inherited cause of intellectual disabilities and a leading cause of autism. FXS is caused by a trinucleotide expansion in the gene FMR1 on the X chromosome. The neuroanatomical hallmark of FXS is an overabundance of immature dendritic spines, a factor thought to underlie synaptic dysfunction and impaired cognition. We showed that aberrantly increased activity of the Rho GTPase Rac1 inhibited the actin-depolymerizing factor cofilin, a major determinant of dendritic spine structure, and caused disease-associated spine abnormalities in the somatosensory cortex of FXS model mice. Increased cofilin phosphorylation and actin polymerization coincided with abnormal dendritic spines and impaired synaptic maturation. Viral delivery of a constitutively active cofilin mutant (cofilinS3A) into the somatosensory cortex of Fmr1-deficient mice rescued the immature dendritic spine phenotype and increased spine density. Inhibition of the Rac1 effector PAK1 with a small-molecule inhibitor rescued cofilin signaling in FXS mice, indicating a causal relationship between PAK1 and cofilin signaling. PAK1 inhibition rescued synaptic signaling (specifically the synaptic ratio of NMDA/AMPA in layer V pyramidal neurons) and improved sensory processing in FXS mice. These findings suggest a causal relationship between increased Rac1-cofilin signaling, synaptic defects, and impaired sensory processing in FXS and uncover a previously unappreciated role for impaired Rac1-cofilin signaling in the aberrant spine morphology and spine density associated with FXS.
Copyright © 2017 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

