Potato starch synthases: Functions and relationships
- PMID: 29114568
- PMCID: PMC5637242
- DOI: 10.1016/j.bbrep.2017.02.004
Potato starch synthases: Functions and relationships
Abstract
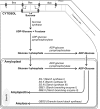
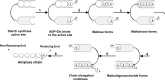
Starch, a very compact form of glucose units, is the most abundant form of storage polyglucan in nature. The starch synthesis pathway is among the central biochemical pathways, however, our understanding of this important pathway regarding genetic elements controlling this pathway, is still insufficient. Starch biosynthesis requires the action of several enzymes. Soluble starch synthases (SSs) are a group of key players in starch biosynthesis which have proven their impact on different aspects of the starch biosynthesis and functionalities. These enzymes have been studied in different plant species and organs in detail, however, there seem to be key differences among species regarding their contributions to the starch synthesis. In this review, we consider an update on various SSs with an emphasis on potato SSs as a model for storage organs. The genetics and regulatory mechanisms of potato starch synthases will be highlighted. Different aspects of various isoforms of SSs are also discussed.
Keywords: Bioinformatics; Glycosyltransferase; Potato; Starch biosynthesis; Starch synthases.
Figures
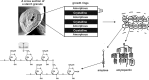
 and
and  represent putative transporters.
represent putative transporters.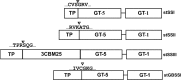

 : represent a glucose moiety with the reducing end.
: represent a glucose moiety with the reducing end.Similar articles
-
Isolation and characterization of cDNAs and genomic DNAs encoding ADP-glucose pyrophosphorylase large and small subunits from sweet potato.Mol Genet Genomics. 2016 Apr;291(2):609-20. doi: 10.1007/s00438-015-1134-3. Epub 2015 Oct 24. Mol Genet Genomics. 2016. PMID: 26499957
-
Genome-wide analysis of starch metabolism genes in potato (Solanum tuberosum L.).BMC Genomics. 2017 Jan 5;18(1):37. doi: 10.1186/s12864-016-3381-z. BMC Genomics. 2017. PMID: 28056783 Free PMC article.
-
Large-scale isolation, fractionation, and purification of soluble starch-synthesizing enzymes: starch synthase and branching enzyme from potato tubers.Carbohydr Res. 2010 Jul 19;345(11):1555-63. doi: 10.1016/j.carres.2010.04.022. Epub 2010 Jun 16. Carbohydr Res. 2010. PMID: 20620253
-
A review of starch-branching enzymes and their role in amylopectin biosynthesis.IUBMB Life. 2014 Aug;66(8):546-58. doi: 10.1002/iub.1297. Epub 2014 Sep 5. IUBMB Life. 2014. PMID: 25196474 Review.
-
Gene expression during tuber development in potato plants.FEBS Lett. 1990 Aug 1;268(2):334-8. doi: 10.1016/0014-5793(90)81281-r. FEBS Lett. 1990. PMID: 2200713 Review.
Cited by
-
Upregulated Lipid Biosynthesis at the Expense of Starch Production in Potato (Solanum tuberosum) Vegetative Tissues via Simultaneous Downregulation of ADP-Glucose Pyrophosphorylase and Sugar Dependent1 Expressions.Front Plant Sci. 2019 Nov 12;10:1444. doi: 10.3389/fpls.2019.01444. eCollection 2019. Front Plant Sci. 2019. PMID: 31781148 Free PMC article.
-
Starch Trek: The Search for Yield.Front Plant Sci. 2019 Jan 21;9:1930. doi: 10.3389/fpls.2018.01930. eCollection 2018. Front Plant Sci. 2019. PMID: 30719029 Free PMC article. Review.
-
Allelic variation in the autotetraploid potato: genes involved in starch and steroidal glycoalkaloid metabolism as a case study.BMC Genomics. 2024 Mar 12;25(1):274. doi: 10.1186/s12864-024-10186-5. BMC Genomics. 2024. PMID: 38475714 Free PMC article.
-
Posttranslational Modification of Waxy to Genetically Improve Starch Quality in Rice Grain.Int J Mol Sci. 2021 May 3;22(9):4845. doi: 10.3390/ijms22094845. Int J Mol Sci. 2021. PMID: 34063649 Free PMC article. Review.
-
Genome-Wide Identification and Expression Analysis of the Starch Synthase Gene Family in Sweet Potato and Two of Its Closely Related Species.Genes (Basel). 2024 Mar 25;15(4):400. doi: 10.3390/genes15040400. Genes (Basel). 2024. PMID: 38674335 Free PMC article.
References
-
- Nazarian-Firouzabadi F., Kok-Jacon G., Vincken J.P., Ji Q., Suurs L.C.J.M., Visser R.G.F. Fusion proteins comprising the catalytic domain of mutansucrase and a starch-binding domain can alter the morphology of amylose-free potato starch granules during biosynthesis. Transgenic Res. 2007;16(5):645–656. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

