Single-Cell Virology: On-Chip Investigation of Viral Infection Dynamics
- PMID: 29117571
- PMCID: PMC5689460
- DOI: 10.1016/j.celrep.2017.10.051
Single-Cell Virology: On-Chip Investigation of Viral Infection Dynamics
Abstract
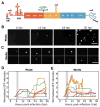
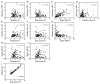
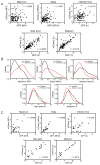
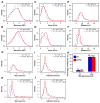
We have developed a high-throughput, microfluidics-based platform to perform kinetic analysis of viral infections in individual cells. We have analyzed thousands of individual poliovirus infections while varying experimental parameters, including multiplicity of infection, cell cycle, viral genotype, and presence of a drug. We make several unexpected observations masked by population-based experiments: (1) viral and cellular factors contribute uniquely and independently to viral infection kinetics; (2) cellular factors cause wide variation in replication start times; and (3) infections frequently begin later and replication occurs faster than predicted by population measurements. We show that mutational load impairs interaction of the viral population with the host, delaying replication start times and explaining the attenuated phenotype of a mutator virus. We show that an antiviral drug can selectively extinguish the most-fit members of the viral population. Single-cell virology facilitates discovery and characterization of virulence determinants and elucidation of mechanisms of drug action eluded by population methods.
Keywords: live-cell imaging; microfluidics; single-cell analysis; stochastic gene expression; temporal dynamics; viral infection; virus-host interaction.
Copyright © 2017 The Authors. Published by Elsevier Inc. All rights reserved.
Figures


References
-
- Appleby TC, Perry JK, Murakami E, Barauskas O, Feng J, Cho A, Fox D, 3rd, Wetmore DR, McGrath ME, Ray AS, et al. Viral replication. Structural basis for RNA replication by the hepatitis C virus polymerase. Science. 2015;347:771–775. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

