Developing selective media for quantification of multispecies biofilms following antibiotic treatment
- PMID: 29121069
- PMCID: PMC5679531
- DOI: 10.1371/journal.pone.0187540
Developing selective media for quantification of multispecies biofilms following antibiotic treatment
Abstract
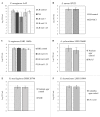
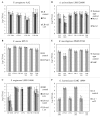
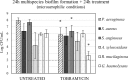
The lungs of cystic fibrosis (CF) patients are chronically colonized by a polymicrobial biofilm community, leading to difficult-to-treat infections. To combat these infections, CF patients are commonly treated with a variety of antibiotics. Understanding the dynamics of polymicrobial community composition in response to antibiotic therapy is essential in the search for novel therapies. Culture-dependent quantification of individual bacteria from defined multispecies biofilms is frequently carried out by plating on selective media. However, the influence of the selective agents in these media on quantitative recovery before or after antibiotic treatment is often unknown. In the present study we developed selective media for six bacterial species that are frequently co-isolated from the CF lung, i.e. Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus anginosus, Achromobacter xylosoxidans, Rothia mucilaginosa, and Gemella haemolysans. We show that certain supplementations to selective media strongly influence quantitative recovery of (un)treated biofilms. Hence, the developed media were optimized for selectivity and quantitative recovery before or after treatment with antibiotics of four major classes, i.e. ceftazidime, ciprofloxacin, colistin, or tobramycin. Finally, in a proof of concept experiment the novel selective media were applied to determine the community composition of multispecies biofilms before and after treatment with tobramycin.
Conflict of interest statement
Figures

Similar articles
-
Antibiotic susceptibility of cystic fibrosis lung microbiome members in a multispecies biofilm.Biofilm. 2020 Jun 13;2:100031. doi: 10.1016/j.bioflm.2020.100031. eCollection 2020 Dec. Biofilm. 2020. PMID: 33447816 Free PMC article.
-
Community Composition Determines Activity of Antibiotics against Multispecies Biofilms.Antimicrob Agents Chemother. 2017 Aug 24;61(9):e00302-17. doi: 10.1128/AAC.00302-17. Print 2017 Sep. Antimicrob Agents Chemother. 2017. PMID: 28696232 Free PMC article.
-
Activity of Antibiotics against Pseudomonas aeruginosa in an In Vitro Model of Biofilms in the Context of Cystic Fibrosis: Influence of the Culture Medium.Antimicrob Agents Chemother. 2020 Mar 24;64(4):e02204-19. doi: 10.1128/AAC.02204-19. Print 2020 Mar 24. Antimicrob Agents Chemother. 2020. PMID: 32015047 Free PMC article.
-
Antibiotic strategies for eradicating Pseudomonas aeruginosa in people with cystic fibrosis.Cochrane Database Syst Rev. 2023 Jun 2;6(6):CD004197. doi: 10.1002/14651858.CD004197.pub6. Cochrane Database Syst Rev. 2023. PMID: 37268599 Free PMC article. Review.
-
Phenazine Antibiotic-Inspired Discovery of Bacterial Biofilm-Eradicating Agents.Chembiochem. 2019 Dec 2;20(23):2885-2902. doi: 10.1002/cbic.201900116. Epub 2019 Oct 2. Chembiochem. 2019. PMID: 30811834 Free PMC article. Review.
Cited by
-
Influence of the lung microbiome on antibiotic susceptibility of cystic fibrosis pathogens.Eur Respir Rev. 2019 Jul 8;28(152):190041. doi: 10.1183/16000617.0041-2019. Print 2019 Jun 30. Eur Respir Rev. 2019. PMID: 31285289 Free PMC article. Review.
-
Examination of a first-in-class bis-dialkylnorspermidine-terphenyl antibiotic in topical formulation against mono and polymicrobial biofilms.PLoS One. 2020 Oct 19;15(10):e0234832. doi: 10.1371/journal.pone.0234832. eCollection 2020. PLoS One. 2020. PMID: 33075071 Free PMC article.
-
Indoor Air Quality and Potential Health Risk Impacts of Exposure to Antibiotic Resistant Bacteria in an Office Rooms in Southern Poland.Int J Environ Res Public Health. 2018 Nov 21;15(11):2604. doi: 10.3390/ijerph15112604. Int J Environ Res Public Health. 2018. PMID: 30469413 Free PMC article.
-
Characterizing species interactions that contribute to biofilm formation in a multispecies model of a potable water bacterial community.Microbiology (Reading). 2020 Jan;166(1):34-43. doi: 10.1099/mic.0.000849. Epub 2019 Oct 4. Microbiology (Reading). 2020. PMID: 31585061 Free PMC article.
-
Microbial diversity and antimicrobial susceptibility in endotracheal tube biofilms recovered from mechanically ventilated COVID-19 patients.Biofilm. 2022 Dec;4:100079. doi: 10.1016/j.bioflm.2022.100079. Epub 2022 Jun 14. Biofilm. 2022. PMID: 35720435 Free PMC article.
References
-
- Lyczak JB, Cannon CL, Pier GB. Lung infections associated with cystic fibrosis. Clin Microbiol Rev. 2002;15(2):194–222. doi: 10.1128/CMR.15.2.194-222.2002 - DOI - PMC - PubMed
-
- Sibley CDSD Surette MGSG. The polymicrobial nature of airway infections in cystic fibrosis: Cangene Gold Medal Lecture. Can J Microbiol. 2011;57(2):69–77. doi: 10.1139/w10-105 - DOI - PubMed
-
- Caverly LJ, Zhao J, LiPuma JJ. Cystic fibrosis lung microbiome: opportunities to reconsider management of airway infection. Pediatr Pulmonol. 2015;50(S40):31–8. - PubMed
-
- de Koff EM, Groot KM de W, Bogaert D. Development of the respiratory tract microbiota in cystic fibrosis. Curr Opin Pulm Med. 2016;22(6):623–8. - PubMed
-
- Huang YJ, LiPuma JJ. The microbiome in cystic fibrosis. Clin Chest Med. 2016;37(1):59–67. doi: 10.1016/j.ccm.2015.10.003 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

