Molecular characterization of a novel Muscovy duck parvovirus isolate: evidence of recombination between classical MDPV and goose parvovirus strains
- PMID: 29121936
- PMCID: PMC5680767
- DOI: 10.1186/s12917-017-1238-6
Molecular characterization of a novel Muscovy duck parvovirus isolate: evidence of recombination between classical MDPV and goose parvovirus strains
Abstract
Background: Muscovy duck parvovirus (MDPV) and Goose parvovirus (GPV) are important etiological agents for Muscovy duck parvoviral disease and Derzsy's disease, respectively; both of which can cause substantial economic losses in waterfowl industry. In contrast to GPV, the complete genomic sequence data of MDPV isolates are still limited and their phylogenetic relationships largely remain unknown. In this study, the entire genome of a pathogenic MDPV strain ZW, which was isolated from a deceased Muscovy duckling in 2006 in China, was cloned, sequenced, and compared with that of other classical MDPV and GPV strains.
Results: The genome of strain ZW comprises of 5071 nucleotides; this genome was shorter than that of the pathogenic MDPV strain YY (5075 nt). All the four deleted nucleotides produced in strain ZW are located at the base-pairing positions in the palindromic stem of inverted terminal repeats (ITR) without influencing the formation of a hairpin structure. Recombination analysis revealed that strain ZW originated from genetic recombination between the classical MDPV and GPV strain. The YY strain of MDPV acts as the major parent, whereas the virulent strains YZ99-6 and B and the vaccine strain SYG61v of GPV act as the minor parents in varying degrees. Two recombination sites were detected in strain ZW, with the small recombination site surrounding the P9 promoter, and the large recombination site situated in the middle of the VP3 gene. The SYG61V strain is a vaccine strain used for preventing goose parvoviral disease. This strain was found to be solely involved in the recombination event detected in the P9 promoter region. Phylogenetic analyses between strain ZW and other classical strains of MDPV and GPV were performed. The results supported the in silico recombination analysis conclusion.
Conclusions: MDPV Strain ZW is a novel recombinant parvovirus, and the bulk of its genome originates from the classical MDPV strain. Two virulent strains and a vaccine strain of GPV were involved in the recombination process in varying degrees.
Keywords: Goose parvovirus; Inverted terminal repeats; Muscovy duck parvovirus; Recombination.
Conflict of interest statement
Ethics approval and consent to participate
The procedure for inoculation of fertilized Muscovy duck eggs was approved by the Animal Care and Use Committee of Yangzhou University and performed in accordance with the “Guidelines for Experimental Animals” of the Ministry of Science and Technology (Beijing, China). No specific permits were required for these locations and activities.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
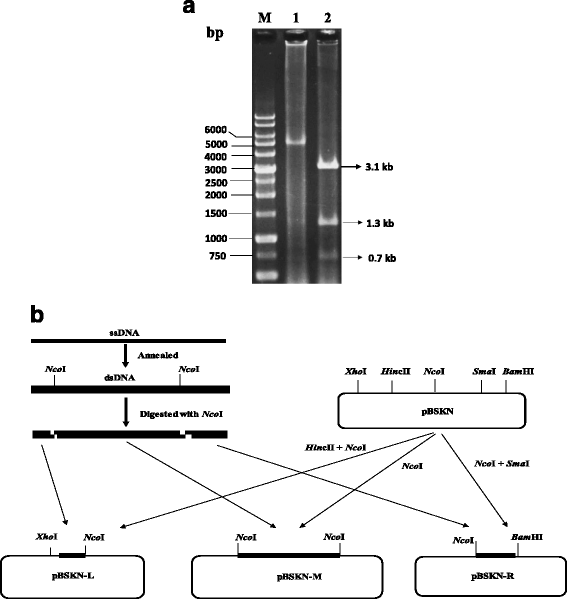

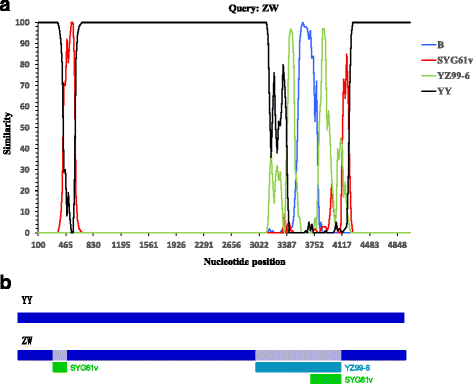
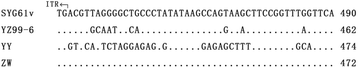
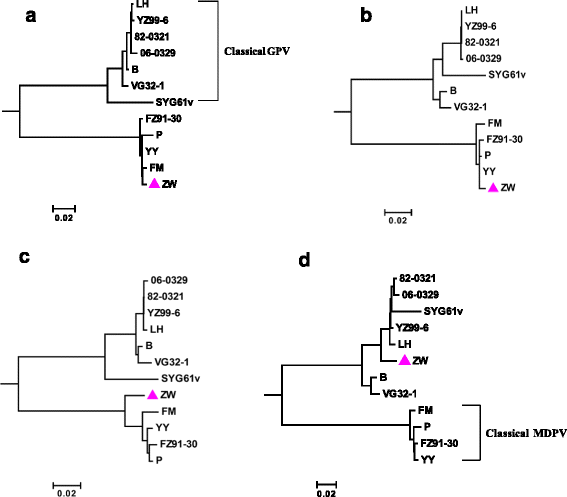
References
-
- Swayne DE, Glisson JR, McDougald LR, Nolan LK, Suarez DL, Nair V. Diseases of poultry (13th edition) Oxford, UK: Wiley-Blackwell Publishing Ltd; 2013. pp. 444–451.
-
- Chen YQ, Lin TL, Hu QL, Li YY, Zhou WM, Wu ZY. Isolation and identification of a Muscovy duckling parvovirus. Chi J Virol. 1993;9(3):228–234.
-
- Jestin V, Le Bras MO, Cherbonnel M, Le Gall G, Bennejean G. Mise en évidence de parvovirus (virus de la maladie de Derzsy) très pathogènes dans les élevages de canards de Barbarie. Rec Med Vet. 1991;167:849–857.
-
- Glávits R, Zolnai A, Szabó É, Ivanics É, Zarka P, Mató T, Palya V. Comparative pathological studies on domestic geese (Anser Anser Domestica) and Muscovy ducks (Cairina Moschata) experimentally infected with parvovirus strains of goose and Muscovy duck origin. Acta Vet Hung. 2005;53:73–89. doi: 10.1556/AVet.53.2005.1.8. - DOI - PubMed
-
- Barnes HJ. Muscovy duck parvovirus. In: Calnek BW, Barnes HJ, Beard CW, McDougald LR, Saif YM, editors. Diseases of poultry. 10. Ames, IA: Iowa State University Press; 1997. pp. 1032–1033.
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

