Chronic cerebral hypoperfusion induces post-stroke dementia following acute ischemic stroke in rats
- PMID: 29121965
- PMCID: PMC5679180
- DOI: 10.1186/s12974-017-0992-5
Chronic cerebral hypoperfusion induces post-stroke dementia following acute ischemic stroke in rats
Abstract
Background: Post-stroke dementia (PSD) is one of the major consequences after stroke. Chronic cerebral hypoperfusion (CCH) can induce vascular cognitive impairment and potentiate amyloid pathology. We investigated how CCH contributes to the development of PSD after stroke in the context of neuroinflammation and amyloid pathology.
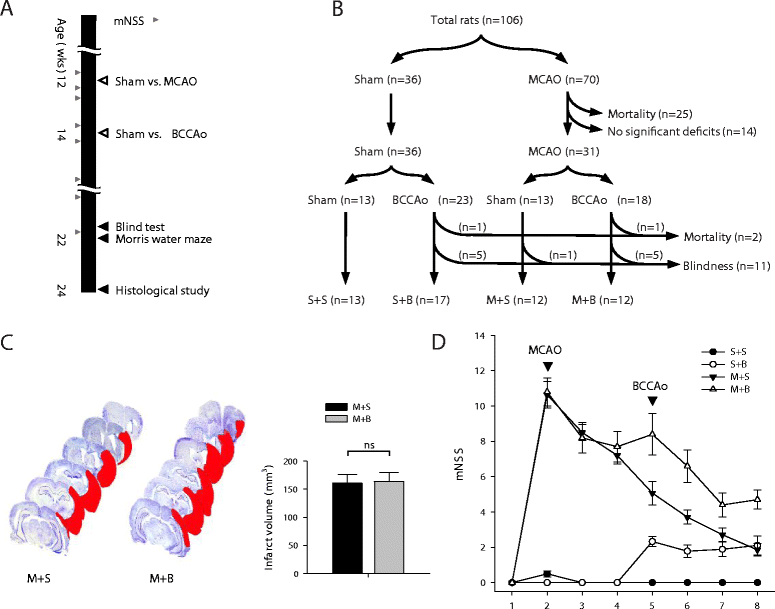
Methods: We designed a unique animal model for PSD. We performed middle cerebral artery occlusion (MCAO) surgery in rats mimicking acute territorial infarct, which was followed by bilateral common carotid artery occlusion (BCCAo) surgery mimicking CCH. We performed behavioral tests including neurologic function test and water maze task and histological investigations including neuroinflammation, neuronal cell death, amyloid pathology, and aquaporin 4 (AQP4) distribution.
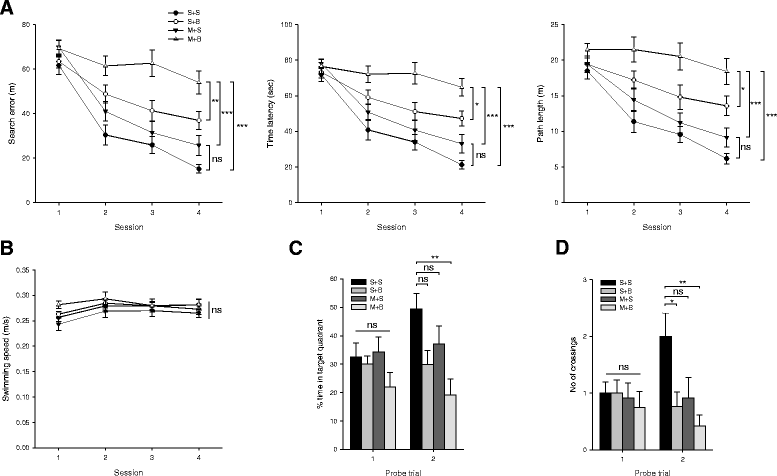
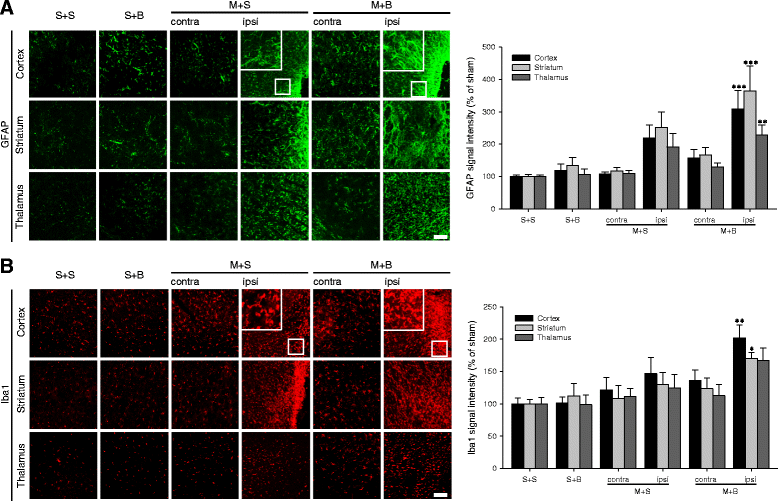
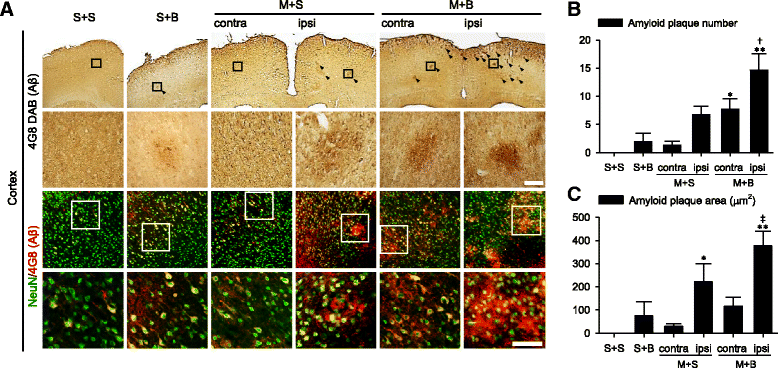
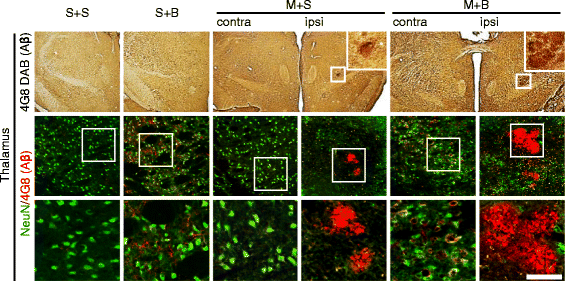
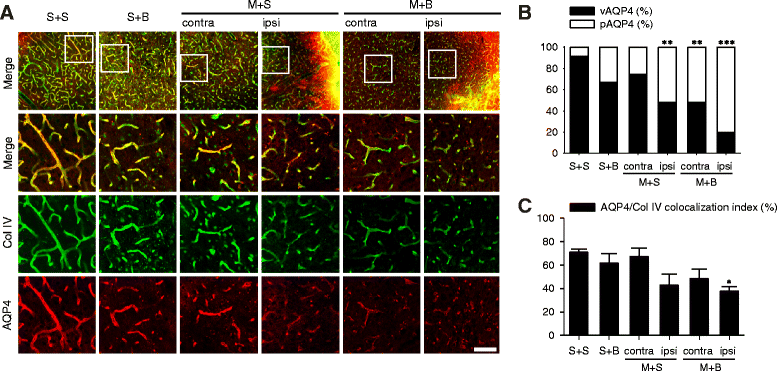
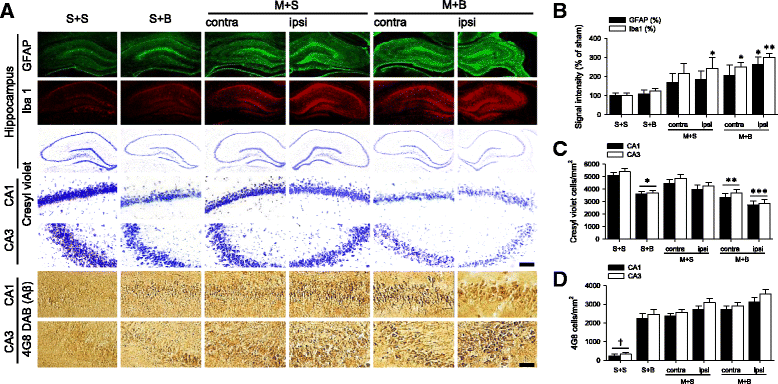
Results: Spatial memory was synergistically impaired when BCCAo was superimposed on MCAO. Neuroinflammation with astroglial or microglial activation and amyloid pathology were enhanced in the ipsilateral cortex, thalamus, and hippocampus when BCCAo was superimposed on MCAO. Glymphatic pathway-related AQP4 distribution changed from perivascular to parenchymal pattern.
Conclusions: Our experimental results suggest that CCH may contribute to the development of PSD by interfering with amyloid clearance through the glymphatic pathway and concomitant neuroinflammation. Therapeutic strategy to clear brain metabolic waste through the glymphatic pathway may be a promising approach to prevent PSD after stroke.
Keywords: Amyloid pathology; Animal model; Chronic cerebral hypoperfusion; Glymphatic pathway; Neuroinflammation; Post-stroke dementia.
Conflict of interest statement
Ethics approval and consent to participate
This study was approved by the Institutional Animal Care and Use Committee of Konkuk University (approval number KU16070).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Characterization of Tauopathy in a Rat Model of Post-Stroke Dementia Combining Acute Infarct and Chronic Cerebral Hypoperfusion.Int J Mol Sci. 2020 Sep 21;21(18):6929. doi: 10.3390/ijms21186929. Int J Mol Sci. 2020. PMID: 32967251 Free PMC article.
-
Synergistic memory impairment through the interaction of chronic cerebral hypoperfusion and amlyloid toxicity in a rat model.Stroke. 2011 Sep;42(9):2595-604. doi: 10.1161/STROKEAHA.111.620179. Epub 2011 Jul 7. Stroke. 2011. PMID: 21737797
-
Efficacy of Artemisia annua Linné in improving cognitive impairment in a chronic cerebral hypoperfusion-induced vascular dementia animal model.Phytomedicine. 2023 Apr;112:154683. doi: 10.1016/j.phymed.2023.154683. Epub 2023 Jan 31. Phytomedicine. 2023. PMID: 36738479
-
Rodent Models of Post-Stroke Dementia.Int J Mol Sci. 2022 Sep 15;23(18):10750. doi: 10.3390/ijms231810750. Int J Mol Sci. 2022. PMID: 36142661 Free PMC article. Review.
-
Molecular Mechanisms of Vascular Dementia: What Can Be Learned from Animal Models of Chronic Cerebral Hypoperfusion?Mol Neurobiol. 2017 Jul;54(5):3670-3682. doi: 10.1007/s12035-016-9915-1. Epub 2016 May 20. Mol Neurobiol. 2017. PMID: 27206432 Review.
Cited by
-
Activation of Neuropeptide Y2 Receptor Can Inhibit Global Cerebral Ischemia-Induced Brain Injury.Neuromolecular Med. 2022 Jun;24(2):97-112. doi: 10.1007/s12017-021-08665-z. Epub 2021 May 21. Neuromolecular Med. 2022. PMID: 34019239 Free PMC article.
-
Protective potential of glucagon like peptide 2 (GLP-2) against the neurodegeneration.Neural Regen Res. 2019 Nov;14(11):1901-1902. doi: 10.4103/1673-5374.259612. Neural Regen Res. 2019. PMID: 31290442 Free PMC article. No abstract available.
-
Role of the glymphatic system and perivascular spaces as a potential biomarker for post-stroke epilepsy.Epilepsia Open. 2024 Feb;9(1):60-76. doi: 10.1002/epi4.12877. Epub 2023 Dec 14. Epilepsia Open. 2024. PMID: 38041607 Free PMC article. Review.
-
The Emerging Roles of Ferroptosis in Vascular Cognitive Impairment.Front Neurosci. 2019 Aug 6;13:811. doi: 10.3389/fnins.2019.00811. eCollection 2019. Front Neurosci. 2019. PMID: 31447633 Free PMC article.
-
Glymphatic dysfunction assessed by DTI-ALPS index predicts early cognitive impairment in acute subcortical infarcts: a prospective clinical cohort study.Front Neurol. 2025 Jul 9;16:1605889. doi: 10.3389/fneur.2025.1605889. eCollection 2025. Front Neurol. 2025. PMID: 40703781 Free PMC article.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

