Sequence analysis and characterization of pyruvate kinase from Clonorchis sinensis, a 53.1-kDa homopentamer, implicated immune protective efficacy against clonorchiasis
- PMID: 29121987
- PMCID: PMC5680780
- DOI: 10.1186/s13071-017-2494-9
Sequence analysis and characterization of pyruvate kinase from Clonorchis sinensis, a 53.1-kDa homopentamer, implicated immune protective efficacy against clonorchiasis
Abstract
Background: Clonorchis sinensis, the causative agent of clonorchiasis, is classified as one of the most neglected tropical diseases and affects more than 15 million people globally. This hepatobiliary disease is highly associated with cholangiocarcinoma. As key molecules in the infectivity and subsistence of trematodes, glycolytic enzymes have been targets for drug and vaccine development. Clonorchis sinensis pyruvate kinase (CsPK), a crucial glycolytic enzyme, was characterized in this research.
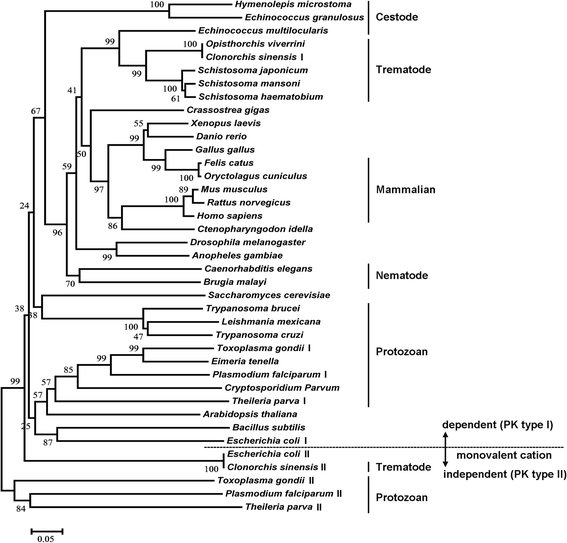
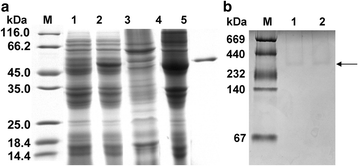
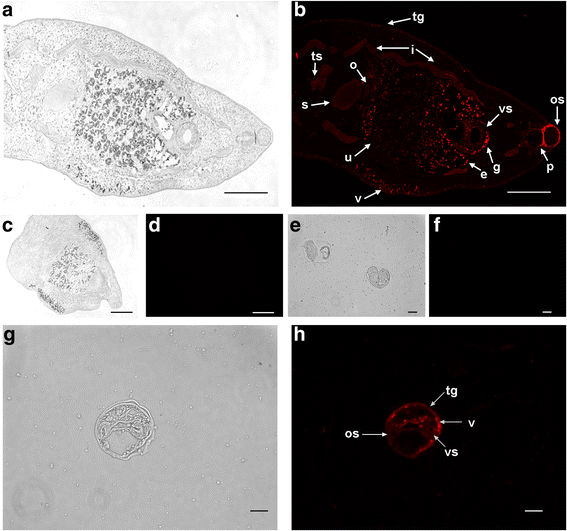
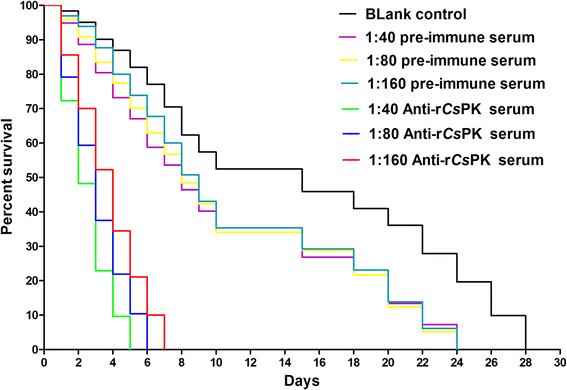
Results: Differences were observed in the sequences and spatial structures of CsPK and PKs from humans, rats, mice and rabbits. CsPK possessed a characteristic active site signature (IKLIAKIENHEGV) and some unique sites but lacked the N-terminal domain. The predicted subunit molecular mass (Mr) of CsPK was 53.1 kDa. Recombinant CsPK (rCsPK) was a homopentamer with a Mr. of approximately 290 kDa by both native PAGE and gel filtration chromatography. Significant differences in the protein and mRNA levels of CsPK were observed among four life stages of C. sinensis (egg, adult worm, excysted metacercaria and metacercaria), suggesting that these developmental stages may be associated with diverse energy demands. CsPK was widely distributed in adult worms. Moreover, an intense Th1-biased immune response was persistently elicited in rats immunized with rCsPK. Also, rat anti-rCsPK sera suppressed C. sinensis adult subsistence both in vivo and in vitro.
Conclusions: The sequences and spatial structures, molecular mass, and expression profile of CsPK have been characterized. rCsPK was indicated to be a homopentamer. Rat anti-rCsPK sera suppressed C. sinensis adult subsistence both in vivo and in vitro. CsPK is worthy of further study as a promising target for drug and vaccine development.
Keywords: Clonorchis sinensis; Drug target; Excretory/secretory products; Expression profile; Immune response; Pentamer; Pyruvate kinase; Vaccine candidate.
Conflict of interest statement
Ethics approval and consent to participate
SD rats and BALB/c mice were purchased from the animal centre of Sun Yat-sen University and raised carefully according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All experimental procedures were authorized by the Animal Care and Use Committee of Sun Yat-sen University (Permit Numbers: SCXK (Guangdong) 2010–0107).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures




Similar articles
-
Molecular identification and characterization of leucine aminopeptidase 2, an excretory-secretory product of Clonorchis sinensis.Mol Biol Rep. 2012 Oct;39(10):9817-26. doi: 10.1007/s11033-012-1848-9. Epub 2012 Jun 23. Mol Biol Rep. 2012. PMID: 22729885
-
Molecular characterization and expression of a cysteine protease from Clonorchis sinensis and its application for serodiagnosis of clonorchiasis.Parasitol Res. 2012 Jun;110(6):2211-9. doi: 10.1007/s00436-011-2751-3. Epub 2011 Dec 15. Parasitol Res. 2012. PMID: 22170263
-
Advanced enzymology, expression profile and immune response of Clonorchis sinensis hexokinase show its application potential for prevention and control of clonorchiasis.PLoS Negl Trop Dis. 2015 Mar 23;9(3):e0003641. doi: 10.1371/journal.pntd.0003641. eCollection 2015 Mar. PLoS Negl Trop Dis. 2015. PMID: 25799453 Free PMC article.
-
Current status and perspectives of Clonorchis sinensis and clonorchiasis: epidemiology, pathogenesis, omics, prevention and control.Infect Dis Poverty. 2016 Jul 6;5(1):71. doi: 10.1186/s40249-016-0166-1. Infect Dis Poverty. 2016. PMID: 27384714 Free PMC article. Review.
-
Clonorchis sinensis and Clonorchiasis: The Relevance of Exploring Genetic Variation.Adv Parasitol. 2018;100:155-208. doi: 10.1016/bs.apar.2018.03.006. Epub 2018 May 4. Adv Parasitol. 2018. PMID: 29753338 Review.
Cited by
-
Molecular Mechanisms of Clonorchis sinensis-Host Interactions and Implications for Vaccine Development.Front Cell Dev Biol. 2022 Jan 18;9:781768. doi: 10.3389/fcell.2021.781768. eCollection 2021. Front Cell Dev Biol. 2022. PMID: 35118069 Free PMC article. Review.
-
Proteomic analysis of Taenia solium cysticercus and adult stages.Front Vet Sci. 2023 Jan 9;9:934197. doi: 10.3389/fvets.2022.934197. eCollection 2022. Front Vet Sci. 2023. PMID: 36699330 Free PMC article.
References
MeSH terms
Substances
Grants and funding
- 2017YFD0501300/the national key research and development program of China
- 2016A050502008/the Science and Technology Planning Project of Guangdong Province
- 16ykpy06/the Young Teachers' Cultivation Project of Basic Scientific Research Service Fee in Colleges and Universities
- 81772212/the National Natural Science Foundation of China
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

