Anticancer Activities of Surfactin and Potential Application of Nanotechnology Assisted Surfactin Delivery
- PMID: 29123482
- PMCID: PMC5662584
- DOI: 10.3389/fphar.2017.00761
Anticancer Activities of Surfactin and Potential Application of Nanotechnology Assisted Surfactin Delivery
Abstract
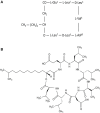
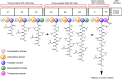
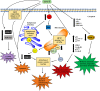
Surfactin, a cyclic lipopeptide biosurfactant produced by various strains of Bacillus genus, has been shown to induce cytotoxicity against many cancer types, such as Ehrlich ascites, breast and colon cancers, leukemia and hepatoma. Surfactin treatment can inhibit cancer progression by growth inhibition, cell cycle arrest, apoptosis, and metastasis arrest. Owing to the potent effect of surfactin on cancer cells, numerous studies have recently investigated the mechanisms that underlie its anticancer activity. The amphiphilic nature of surfactin allows its easy incorporation nano-formulations, such as polymeric nanoparticles, micelles, microemulsions, liposomes, to name a few. The use of nano-formulations offers the advantage of optimizing surfactin delivery for an improved anticancer therapy. This review focuses on the current knowledge of surfactin properties and biosynthesis; anticancer activity against different cancer models and the underlying mechanisms involved; as well as the potential application of nano-formulations for optimal surfactin delivery.
Keywords: anticancer; biosurfactant; lipopeptide; nano-formulation; surfactin.
Figures



Similar articles
-
Toxicity and applications of surfactin for health and environmental biotechnology.J Toxicol Environ Health B Crit Rev. 2018;21(6-8):382-399. doi: 10.1080/10937404.2018.1564712. Epub 2019 Jan 7. J Toxicol Environ Health B Crit Rev. 2018. PMID: 30614421 Review.
-
Sustainable Production of Biosurfactant from Agro-Industrial Oil Wastes by Bacillus subtilis and Its Potential Application as Antioxidant and ACE Inhibitor.Int J Mol Sci. 2022 Sep 16;23(18):10824. doi: 10.3390/ijms231810824. Int J Mol Sci. 2022. PMID: 36142732 Free PMC article.
-
The Surfactin-Like Lipopeptides From Bacillus spp.: Natural Biodiversity and Synthetic Biology for a Broader Application Range.Front Bioeng Biotechnol. 2021 Mar 2;9:623701. doi: 10.3389/fbioe.2021.623701. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 33738277 Free PMC article.
-
Surfactin as an ingredient in cosmetic industry: Benefits and trends.Int J Cosmet Sci. 2024 Oct;46(5):702-716. doi: 10.1111/ics.12957. Epub 2024 Mar 13. Int J Cosmet Sci. 2024. PMID: 38481065 Review.
-
Cell-free biosynthesis of surfactin, a cyclic lipopeptide produced by Bacillus subtilis.Biochemistry. 1991 Jul 2;30(26):6503-8. doi: 10.1021/bi00240a022. Biochemistry. 1991. PMID: 1905154
Cited by
-
Lichenysin Production by Bacillus licheniformis Food Isolates and Toxicity to Human Cells.Front Microbiol. 2022 Feb 7;13:831033. doi: 10.3389/fmicb.2022.831033. eCollection 2022. Front Microbiol. 2022. PMID: 35197958 Free PMC article.
-
Harnessing the Potential of Biosurfactants for Biomedical and Pharmaceutical Applications.Pharmaceutics. 2023 Aug 18;15(8):2156. doi: 10.3390/pharmaceutics15082156. Pharmaceutics. 2023. PMID: 37631370 Free PMC article. Review.
-
Molecular characterization and in silico evaluation of surfactins produced by endophytic bacteria from Phanera splendens.Front Chem. 2023 Aug 7;11:1240704. doi: 10.3389/fchem.2023.1240704. eCollection 2023. Front Chem. 2023. PMID: 37608862 Free PMC article.
-
Complete genome sequence and antimicrobial activity of Bacillus velezensis JT3-1, a microbial germicide isolated from yak feces.3 Biotech. 2020 May;10(5):231. doi: 10.1007/s13205-020-02235-z. Epub 2020 May 5. 3 Biotech. 2020. PMID: 32399381 Free PMC article.
-
Production, characterization and biomedical potential of biosurfactants produced by haloalkaliphilic archaea from Wadi El-Natrun, Egypt.Microb Cell Fact. 2024 Mar 14;23(1):84. doi: 10.1186/s12934-024-02351-y. Microb Cell Fact. 2024. PMID: 38486239 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

