Comparison of the efficacy of continuous i.v. infusion versus continuous regional arterial infusion of nafamostat mesylate for severe acute pancreatitis
- PMID: 29123791
- PMCID: PMC5667240
- DOI: 10.1002/ams2.173
Comparison of the efficacy of continuous i.v. infusion versus continuous regional arterial infusion of nafamostat mesylate for severe acute pancreatitis
Abstract
Aim: Continuous regional arterial infusion (CRAI) of protease inhibitors may be effective in the treatment of severe acute pancreatitis (SAP), but it is more invasive than i.v. infusion. The purpose of this study was to examine the effectiveness of continuous i.v. infusion (CIVI) for SAP compared with CRAI by unifying the dose and the administration period of nafamostat mesylate.
Methods: This study comprised 32 patients with SAP who were divided into two groups: the CRAI group and the CIVI group. The protease inhibitor, nafamostat mesylate, was continuously infused at a rate of 200 mg/day for 5 days in both groups. Clinical outcomes including in-hospital mortality were examined.
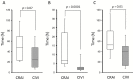
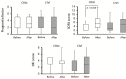
Results: There were no significant between-group differences in in-hospital mortality and 90-day mortality. The duration from admission to treatment was significantly shorter in the CIVI group (median, 7 h vs. 2 h, P = 0.0001; CRAI group vs. CIVI group). The rate of mechanical ventilation was significantly less in the CIVI group than in the CRAI group (93% vs. 47%, P = 0.007). The CIVI group showed a tendency toward decreased length of intensive care unit stay (median, 13 days vs. 4 days, P = 0.085) and hospital stay (median, 19 days vs. 11 days, P = 0.072). Total costs during hospitalization were significantly lower in the CIVI group (median, $18,320 vs. $11,641, P = 0.049).
Conclusion: The effectiveness of CIVI with early nafamostat mesylate treatment after the development of SAP could be equivalent to, or better than, that of CRAI.
Keywords: Continuous intravenous infusion; continuous regional arterial infusion; nafamostat mesylate; protease inhibitor; severe acute pancreatitis.
Figures
References
-
- Niederau C, Liddle RA, Ferrell LD, Grendell JH. Beneficial effects of cholecystokinin‐receptor blockade and inhibition of proteolytic enzyme activity in experimental acute hemorrhagic pancreatitis in mice. Evidence for cholecystokinin as a major factor in the development of acute pancreatitis. J. Clin. Invest. 1986; 78: 1056–1063. - PMC - PubMed
-
- Lankisch PG, Pohl U, Göke B et al Effect of FOY‐305 (camostat) on severe acute pancreatitis in two experimental animal models. Gastroenterology 1989; 96: 193–199. - PubMed
-
- Suzuki M, Isaji S, Stanten R, Frey CF, Ruebner B. Effect of protease inhibitor FUT‐175 on acute hemorrhagic pancreatitis in mice. Int. J. Pancreatol. 1992; 11: 578–582. - PubMed
-
- Takeda K. Antiproteases in the treatment of acute necrotizing pancreatitis: Continuous regional arterial infusion. JOP 2007; 8 (Suppl. 4): 526–532. - PubMed
-
- Luh SP, Tsai CC, Shau WY et al Effects of gabexate mesilate (FOY) on ischemia‐reperfusion‐induced acute lung injury in dogs. J. Surg. Res. 1999; 87: 152–163. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

