Alteration of Gut Microbiota and Inflammatory Cytokine/Chemokine Profiles in 5-Fluorouracil Induced Intestinal Mucositis
- PMID: 29124041
- PMCID: PMC5662589
- DOI: 10.3389/fcimb.2017.00455
Alteration of Gut Microbiota and Inflammatory Cytokine/Chemokine Profiles in 5-Fluorouracil Induced Intestinal Mucositis
Abstract
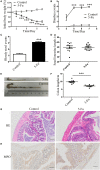
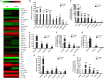
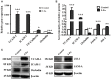
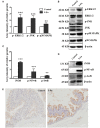
Disturbed homeostasis of gut microbiota has been suggested to be closely associated with 5-fluorouracil (5-Fu) induced mucositis. However, current knowledge of the overall profiles of 5-Fu-disturbed gut microbiota is limited, and so far there is no direct convincing evidence proving the causality between 5-Fu-disturbed microbiota and colonic mucositis. In mice, in agreement with previous reports, 5-Fu resulted in severe colonic mucositis indicated by weight loss, diarrhea, bloody stool, shortened colon, and infiltration of inflammatory cells. It significantly changed the profiles of inflammatory cytokines/chemokines in serum and colon. Adhesion molecules such as vascular cell adhesion molecule-1 (VCAM-1), intercellular adhesion molecule-1 (ICAM-1), and VE-Cadherin were increased. While tight junction protein occludin was reduced, however, zonula occludens-1 (ZO-1) and junctional adhesion molecule-A (JAM-A) were increased in colonic tissues of 5-Fu treated mice. Meanwhile, inflammation related signaling pathways including NF-κB and mitogen activated protein kinase (MAPKs) in the colon were activated. Further study disclosed that 5-Fu diminished bacterial community richness and diversity, leading to the relative lower abundance of Firmicutes and decreased Firmicutes/Bacteroidetes (F/B) ratio in feces and cecum contents. 5-Fu also reduced the proportion of Proteobacteria, Tenericutes, Cyanobacteria, and Candidate division TM7, but increased that of Verrucomicrobia and Actinobacteria in feces and/or cecum contents. The fecal transplant from healthy mice prevented body weight loss and colon shortening of 5-Fu treated mice. In addition, the fecal transplant from 5-Fu treated mice reduced body weight and colon length of vancomycin-pretreated mice. Taken together, our study demonstrated that gut microbiota was actively involved in the pathological process of 5-Fu induced intestinal mucositis, suggesting potential attenuation of 5-Fu induced intestinal mucositis by manipulating gut microbiota homeostasis.
Keywords: 5-fluorouracil; fecal transplantation; gut microbiota; inflammatory chemokines/cytokines; intestinal mucositis.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

