Association of lamin A/C with muscle gene-specific promoters in myoblasts
- PMID: 29124189
- PMCID: PMC5668900
- DOI: 10.1016/j.bbrep.2015.08.021
Association of lamin A/C with muscle gene-specific promoters in myoblasts
Abstract
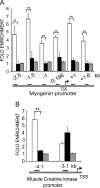
The A-type and B-type lamins form a filamentous meshwork underneath the inner nuclear membrane called the nuclear lamina, which is an important component of nuclear architecture in metazoan cells. The lamina interacts with large, mostly repressive chromatin domains at the nuclear periphery. In addition, genome-lamina interactions also involve dynamic association of lamin A/C with gene promoters in adipocytes. Mutations in the human lamin A gene cause a spectrum of hereditary diseases called the laminopathies which affect muscle, cardiac and adipose tissues. Since most mutations in lamin A/C affect skeletal muscle, we investigated lamin-chromatin interactions at promoters of muscle specific genes in both muscle and non-muscle cell lines by ChIP-qPCR. We observed that lamin A/C was specifically associated with promoter regions of muscle genes in myoblasts but not in fibroblasts. Lamin A/C dissociated from the promoter regions of the differentiation specific MyoD, myogenin and muscle creatine kinase genes when myoblasts were induced to differentiate. In the promoter regions of the myogenin and MyoD genes, the binding of lamin A/C in myoblasts inversely correlated with the active histone mark, H3K4me3. Lamin A/C binding on muscle genes was reduced and differentiation potential was enhanced on treatment of myoblasts with a histone deacetylase inhibitor. These findings suggest a role for lamina-chromatin interactions in muscle differentiation and have important implications for the pathological mechanisms of striated muscle associated laminopathies.
Keywords: Genome–lamina interactions; Laminopathies; Lamins; Muscle differentiation; Myogenin; Nuclear lamina.
Figures



Similar articles
-
Protein Kinase C Alpha Cellular Distribution, Activity, and Proximity with Lamin A/C in Striated Muscle Laminopathies.Cells. 2020 Oct 31;9(11):2388. doi: 10.3390/cells9112388. Cells. 2020. PMID: 33142761 Free PMC article.
-
Laminopathy-causing lamin A mutations reconfigure lamina-associated domains and local spatial chromatin conformation.Nucleus. 2018 Jan 1;9(1):216-226. doi: 10.1080/19491034.2018.1449498. Nucleus. 2018. PMID: 29517398 Free PMC article. Review.
-
Nuclear lamins: making contacts with promoters.Nucleus. 2013 Nov-Dec;4(6):424-30. doi: 10.4161/nucl.26865. Epub 2013 Nov 8. Nucleus. 2013. PMID: 24213377 Free PMC article.
-
Expression of the myodystrophic R453W mutation of lamin A in C2C12 myoblasts causes promoter-specific and global epigenetic defects.Exp Cell Res. 2008 May 1;314(8):1869-80. doi: 10.1016/j.yexcr.2008.02.018. Epub 2008 Mar 7. Exp Cell Res. 2008. PMID: 18396274
-
Lamin A, Chromatin and FPLD2: Not Just a Peripheral Ménage-à-Trois.Front Cell Dev Biol. 2018 Jul 9;6:73. doi: 10.3389/fcell.2018.00073. eCollection 2018. Front Cell Dev Biol. 2018. PMID: 30057899 Free PMC article. Review.
Cited by
-
PCAF Involvement in Lamin A/C-HDAC2 Interplay during the Early Phase of Muscle Differentiation.Cells. 2020 Jul 20;9(7):1735. doi: 10.3390/cells9071735. Cells. 2020. PMID: 32698523 Free PMC article.
-
Altered modulation of lamin A/C-HDAC2 interaction and p21 expression during oxidative stress response in HGPS.Aging Cell. 2018 Oct;17(5):e12824. doi: 10.1111/acel.12824. Epub 2018 Aug 15. Aging Cell. 2018. PMID: 30109767 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

