Nitric oxide production by glomerular podocytes
- PMID: 29128399
- PMCID: PMC5776695
- DOI: 10.1016/j.niox.2017.11.005
Nitric oxide production by glomerular podocytes
Abstract
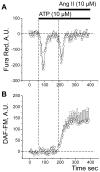
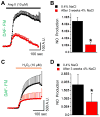
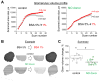
Nitric Oxide (NO), a potent vasodilator and vital signaling molecule, has been shown to contribute to the regulation of glomerular ultrafiltration. However, whether changes in NO occur in podocytes during the pathogenesis of salt-sensitive hypertension has not yet been thoroughly examined. We showed here that podocytes produce NO, and further hypothesized that hypertensive animals would exhibit reduced NO production in these cells in response to various paracrine factors, which might contribute to the damage of glomeruli filtration barrier and development of proteinuria. To test this, we isolated glomeruli from the kidneys of Dahl salt-sensitive (SS) rats fed a low salt (LS; 0.4% NaCl) or high salt (HS; 4% NaCl, 3 weeks) diets and loaded podocytes with either a combination of NO and Ca2+ fluorophores (DAF-FM and Fura Red, respectively) or DAF-FM alone. Changes in fluorescence were observed with confocal microscopy in response to adenosine triphosphate (ATP), angiotensin II (Ang II), and hydrogen peroxide (H2O2). Application of Ang II resulted in activation of both NO and intracellular calcium ([Ca2+]i) transients. In contrast, ATP promoted [Ca2+]i transients, but did not have any effects on NO production. SS rats fed a HS diet for 3 weeks demonstrated impaired NO production: the response to Ang II or H2O2 in podocytes of glomeruli isolated from SS rats fed a HS diet was significantly reduced compared to rats fed a LS diet. Therefore, glomerular podocytes from hypertensive rats showed a diminished NO release in response to Ang II or oxidative stress, suggesting that podocytic NO signaling is dysfunctional in this condition and likely contributes to the development of kidney injury.
Keywords: Angiotensin II; DAF-FM; Dahl salt-sensitive rat; Hydrogen peroxide; Hypertension; Nitric oxide.
Copyright © 2017 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare that they have no conflict of interest related to the publication of this manuscript.
Figures


Similar articles
-
Reduced angiotensin II and oxidative stress contribute to impaired vasodilation in Dahl salt-sensitive rats on low-salt diet.Hypertension. 2005 Apr;45(4):687-91. doi: 10.1161/01.HYP.0000154684.40599.03. Epub 2005 Feb 14. Hypertension. 2005. PMID: 15710779
-
A NOX4/TRPC6 Pathway in Podocyte Calcium Regulation and Renal Damage in Diabetic Kidney Disease.J Am Soc Nephrol. 2018 Jul;29(7):1917-1927. doi: 10.1681/ASN.2018030280. Epub 2018 May 23. J Am Soc Nephrol. 2018. PMID: 29793963 Free PMC article.
-
Podocyte injury underlies the glomerulopathy of Dahl salt-hypertensive rats and is reversed by aldosterone blocker.Hypertension. 2006 Jun;47(6):1084-93. doi: 10.1161/01.HYP.0000222003.28517.99. Epub 2006 Apr 24. Hypertension. 2006. PMID: 16636193
-
Salt-induced hemodynamic regulation mediated by nitric oxide.J Hypertens. 2011 Mar;29(3):415-24. doi: 10.1097/HJH.0b013e328341d19e. J Hypertens. 2011. PMID: 21150639 Review.
-
Role of the adenosine(2A) receptor-epoxyeicosatrienoic acid pathway in the development of salt-sensitive hypertension.Prostaglandins Other Lipid Mediat. 2012 Aug;98(3-4):39-47. doi: 10.1016/j.prostaglandins.2011.12.002. Epub 2011 Dec 22. Prostaglandins Other Lipid Mediat. 2012. PMID: 22227265 Free PMC article. Review.
Cited by
-
Redox regulation in diabetic kidney disease.Am J Physiol Renal Physiol. 2023 Aug 1;325(2):F135-F149. doi: 10.1152/ajprenal.00047.2023. Epub 2023 Jun 1. Am J Physiol Renal Physiol. 2023. PMID: 37262088 Free PMC article. Review.
-
Nitric-Oxide-Mediated Signaling in Podocyte Pathophysiology.Biomolecules. 2022 May 25;12(6):745. doi: 10.3390/biom12060745. Biomolecules. 2022. PMID: 35740870 Free PMC article. Review.
-
The Life of a Kidney Podocyte.Acta Physiol (Oxf). 2025 Aug;241(8):e70081. doi: 10.1111/apha.70081. Acta Physiol (Oxf). 2025. PMID: 40698593 Free PMC article. Review.
-
β-Arrestin pathway activation by selective ATR1 agonism promotes calcium influx in podocytes, leading to glomerular damage.Clin Sci (Lond). 2023 Dec 22;137(24):1789-1804. doi: 10.1042/CS20230313. Clin Sci (Lond). 2023. PMID: 38051199 Free PMC article.
-
Repurposing Riociguat to Target a Novel Paracrine Nitric Oxide-TRPC6 Pathway to Prevent Podocyte Injury.Int J Mol Sci. 2021 Nov 19;22(22):12485. doi: 10.3390/ijms222212485. Int J Mol Sci. 2021. PMID: 34830371 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

