Inborn errors of immunity underlying fungal diseases in otherwise healthy individuals
- PMID: 29128761
- PMCID: PMC5733726
- DOI: 10.1016/j.mib.2017.10.016
Inborn errors of immunity underlying fungal diseases in otherwise healthy individuals
Abstract
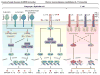
It has been estimated that there are at least 1.5 million fungal species, mostly present in the environment, but only a few of these fungi cause human disease. Most fungal diseases are self-healing and benign, but some are chronic or life-threatening. Acquired and inherited defects of immunity, including breaches of mucocutaneous barriers and circulating leukocyte deficiencies, account for most severe modern-day mycoses. Other types of infection typically accompany these fungal infections. More rarely, severe fungal diseases can strike otherwise healthy individuals. Historical reports of fungi causing chronic peripheral infections (e.g. affecting the nails, skin, hair), and invasive diseases (e.g. brain, lungs, liver), in otherwise healthy patients, can be traced back to the mid-20th century. These fungi typically cause endemic, but not epidemic diseases, are more likely to underlie sporadic than familial cases, and only threaten a small proportion of infected individuals. The basis of this 'idiosyncratic' susceptibility has long remained unexplained, but it has recently become apparent that 'idiopathic' fungal diseases, in children, teenagers, and even adults, may be caused by single-gene inborn errors of immunity. The study of these unusual primary immunodeficiencies (PIDs) has led to the identification of molecules and cells playing a crucial role in human host defenses against certain fungi at particular anatomic sites. A picture is emerging of inborn errors of IL-17 immunity selectively underlying chronic mucocutaneous candidiasis, with little inter-individual variability, and of inborn errors of CARD9 immunity underlying various life-threatening invasive fungal diseases, differing between patients.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures

References
-
- Brown GD, Denning DW, Gow NA, Levitz SM, Netea MG, White TC. Hidden killers: human fungal infections. Sci Transl Med. 2012;4:165rv113. - PubMed
-
- Chakrabarti A, Singh R. The emerging epidemiology of mould infections in developing countries. Curr Opin Infect Dis. 2011;24:521–526. - PubMed
-
- Lass-Florl C. The changing face of epidemiology of invasive fungal disease in Europe. Mycoses. 2009;52:197–205. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

