Tripodal Amine Ligands for Accelerating Cu-Catalyzed Azide-Alkyne Cycloaddition: Efficiency and Stability against Oxidation and Dissociation
- PMID: 29129990
- PMCID: PMC5679428
- DOI: 10.1039/C7CY00587C
Tripodal Amine Ligands for Accelerating Cu-Catalyzed Azide-Alkyne Cycloaddition: Efficiency and Stability against Oxidation and Dissociation
Abstract
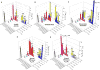
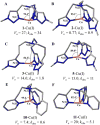
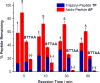
Ancillary ligands, especially the tripodal ligands such as tris(triazolylmethyl)amines, have been widely used to accelerate the Cu-catalyzed azide-alkyne cycloaddition (CuAAC, a "click" reaction). However, the relationship between the activity of these Cu(I) complexes and their stability against air oxidation and ligand dissociation/exchange was seldom studied, which is critical for the applications of CuAAC in many biological systems. In this work, we synthesized twenty-one Cu(I) tripodal ligands varying in chelate arm length (five to seven atoms), donor groups (triazolyl, pyridyl and phenyl), and steric hindrance. The effects of these variables on the CuAAC reaction, air oxidation, and ligand dissociation were evaluated. Reducing the chelate arm length to five atoms, decreasing steric hindrance, or using a relatively weakly-binding ligand can significantly increase the CuAAC reactivity of the Cu(I) complexes, but the concomitant higher degree of oxidation cannot be avoided, which leads to rapid degradation of a histidine-containing peptide as a model of proteins. The oxidation of the peptide can be reduced by attaching oligo(ethylene glycol) chains to the ligands as sacrificing reagents. Using electrospray ionization mass spectrometry (ESI-MS), we directly observed the tri- and di-copper(I)-acetylide complexes in CuAAC reaction in the [5,5,5] ligand system and a small amount of di-Cu(I)-acetylide in the [5,5,6] ligand system. Only the mono-Cu(I) ligand adducts were observed in the [6,6,6] and [5,6,6] ligand systems.
Keywords: ascorbic acid; copper(I) ligands; copper-catalyzed alkyne-azide cycloaddition; dissociation constant; oxidation.
Figures



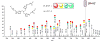

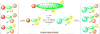
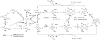
Similar articles
-
Intermediates Stabilized by Tris(triazolylmethyl)amines in the CuAAC Reaction.Chemistry. 2017 Apr 3;23(19):4730-4735. doi: 10.1002/chem.201700555. Epub 2017 Mar 15. Chemistry. 2017. PMID: 28191741
-
Copper and silver complexes of tris(triazole)amine and tris(benzimidazole)amine ligands: evidence that catalysis of an azide-alkyne cycloaddition ("click") reaction by a silver tris(triazole)amine complex arises from copper impurities.Inorg Chem. 2014 Jul 7;53(13):6503-11. doi: 10.1021/ic5008999. Epub 2014 Jun 20. Inorg Chem. 2014. PMID: 24949519
-
Azide-Alkyne "Click" Reaction in Water Using Parts-Per-Million Amine-Functionalized Azoaromatic Cu(I) Complex as Catalyst: Effect of the Amine Side Arm.Inorg Chem. 2021 Dec 6;60(23):17537-17554. doi: 10.1021/acs.inorgchem.1c02115. Epub 2021 Nov 21. Inorg Chem. 2021. PMID: 34806366
-
In-situ Generated and Premade 1-Copper(I) Alkynes in Cycloadditions.Chem Rec. 2017 Dec;17(12):1231-1248. doi: 10.1002/tcr.201700011. Epub 2017 Jun 22. Chem Rec. 2017. PMID: 28639363 Review.
-
Click dendrimers and triazole-related aspects: catalysts, mechanism, synthesis, and functions. A bridge between dendritic architectures and nanomaterials.Acc Chem Res. 2012 Apr 17;45(4):630-40. doi: 10.1021/ar200235m. Epub 2011 Dec 8. Acc Chem Res. 2012. PMID: 22148925 Review.
Cited by
-
Macrocyclization strategies for cyclic peptides and peptidomimetics.RSC Med Chem. 2021 Jun 29;12(8):1325-1351. doi: 10.1039/d1md00083g. eCollection 2021 Aug 18. RSC Med Chem. 2021. PMID: 34447937 Free PMC article. Review.
-
Copper Ligand and Anion Effects: Controlling the Kinetics of the Photoinitiated Copper(I) Catalyzed Azide-Alkyne Cycloaddition Polymerization.Polym Chem. 2018 Oct 14;9(38):4772-4780. doi: 10.1039/C8PY01004H. Epub 2018 Aug 24. Polym Chem. 2018. PMID: 31031838 Free PMC article.
References
-
- Tornøe CW, Christensen C, Meldal M. J. Org. Chem. 2002;67:3057–3064. - PubMed
-
- Rostovtsev VV, Green LG, Fokin VV, Sharpless KB. Angew. Chem. Int. Ed. 2002;41:2596–2599. - PubMed
-
- Kolb HC, Finn M, Sharpless KB. Angew. Chem. Int. Ed. 2001;40:2004–2021. - PubMed
-
- Tron GC, Pirali T, Billington RA, Canonico PL, Sorba G, Genazzani AA. Med. Res. Rev. 2008;28:278–308. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
