Acupuncture for postprandial distress syndrome (APDS): study protocol for a randomized controlled trial
- PMID: 29132415
- PMCID: PMC5683366
- DOI: 10.1186/s13063-017-2285-9
Acupuncture for postprandial distress syndrome (APDS): study protocol for a randomized controlled trial
Abstract
Background: Postprandial distress syndrome (PDS) is referred to as meal-related functional dyspepsia (FD) and causes a reduced quality of life (QoL) for patients. Several randomized controlled trials (RCTs) have suggested that acupuncture is an effective treatment for FD, but few studies were particularly for PDS. This pilot study was designed to determine the feasibility and efficacy of acupuncture in patients with PDS characterized by postprandial fullness and early satiation according to the Rome III criteria.
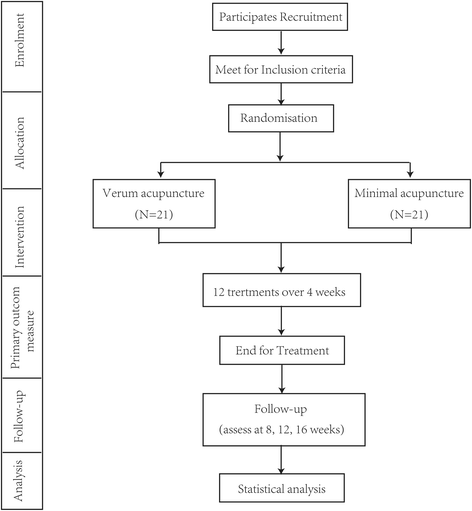
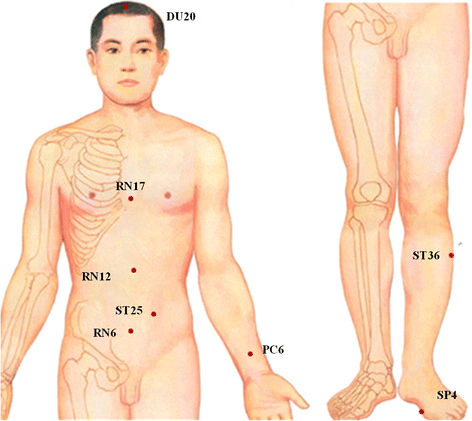
Methods: This is a multi-center, two-arm, blinded (participants), pilot RCT. Forty-two participants who meet the inclusion criteria will be randomly assigned to the verum acupuncture group or minimal acupuncture group in a 1:1 ratio. Both treatments consist of 12 sessions of 20 min duration over four weeks (three sessions per week). The primary outcome measurement is the proportion of persons who improve as assessed using the global outcome by the overall treatment effect (OTE) at end-of-treatment (EOT) (four weeks after randomization). Global assessment at weeks 8 and 16 after randomization is one of the secondary outcomes. The other secondary outcomes including symptoms, disease-specific QoL, and depression and anxiety will be assessed at weeks 4, 8, and 16 after randomization.
Discussion: This pilot study will help determine the feasibility and efficacy of acupuncture in patients with PDS.
Trial registration: ISRCTN Registry, ISRCTN18135146 . Registered on 7 July 2016.
Keywords: Acupuncture; Functional dyspepsia; Minimal acupuncture; Postprandial distress syndrome; Randomized controlled trial.
Conflict of interest statement
Ethics approval and consent to participate
The central ethics committee Research Ethical Committee of Beijing Hospital of Traditional Chinese Medicine Affiliated to Capital Medical University has approved the study protocol in all the centers (reference: 2016BL-011-01). Before randomization, all participants will be requested to provide written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous