Force Spectroscopy with 9-μs Resolution and Sub-pN Stability by Tailoring AFM Cantilever Geometry
- PMID: 29132641
- PMCID: PMC5770970
- DOI: 10.1016/j.bpj.2017.10.023
Force Spectroscopy with 9-μs Resolution and Sub-pN Stability by Tailoring AFM Cantilever Geometry
Abstract
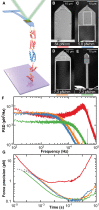
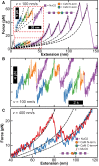
Atomic force microscopy (AFM)-based single-molecule force spectroscopy (SMFS) is a powerful yet accessible means to characterize the unfolding/refolding dynamics of individual molecules and resolve closely spaced, transiently occupied folding intermediates. On a modern commercial AFM, these applications and others are now limited by the mechanical properties of the cantilever. Specifically, AFM-based SMFS data quality is degraded by a commercial cantilever's limited combination of temporal resolution, force precision, and force stability. Recently, we modified commercial cantilevers with a focused ion beam to optimize their properties for SMFS. Here, we extend this capability by modifying a 40 × 18 μm2 cantilever into one terminated with a gold-coated, 4 × 4 μm2 reflective region connected to an uncoated 2-μm-wide central shaft. This "Warhammer" geometry achieved 8.5-μs resolution coupled with improved force precision and sub-pN stability over 100 s when measured on a commercial AFM. We highlighted this cantilever's biological utility by first resolving a calmodulin unfolding intermediate previously undetected by AFM and then measuring the stabilization of calmodulin by myosin light chain kinase at dramatically higher unfolding velocities than in previous AFM studies. More generally, enhancing data quality via an improved combination of time resolution, force precision, and force stability will broadly benefit biological applications of AFM.
Published by Elsevier Inc.
Figures

Comment in
-
Warhammers for Peaceful Times.Biophys J. 2018 Jan 9;114(1):1-2. doi: 10.1016/j.bpj.2017.10.043. Biophys J. 2018. PMID: 29320677 Free PMC article.
References
-
- Liphardt J., Onoa B., Bustamante C. Reversible unfolding of single RNA molecules by mechanical force. Science. 2001;292:733–737. - PubMed
-
- Rief M., Gautel M., Gaub H.E. Reversible unfolding of individual titin immunoglobulin domains by AFM. Science. 1997;276:1109–1112. - PubMed
-
- Cecconi C., Shank E.A., Marqusee S. Direct observation of the three-state folding of a single protein molecule. Science. 2005;309:2057–2060. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

