Recovery of taste organs and sensory function after severe loss from Hedgehog/Smoothened inhibition with cancer drug sonidegib
- PMID: 29133390
- PMCID: PMC5715770
- DOI: 10.1073/pnas.1712881114
Recovery of taste organs and sensory function after severe loss from Hedgehog/Smoothened inhibition with cancer drug sonidegib
Abstract
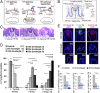
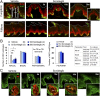
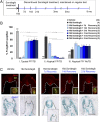
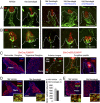
Striking taste disturbances are reported in cancer patients treated with Hedgehog (HH)-pathway inhibitor drugs, including sonidegib (LDE225), which block the HH pathway effector Smoothened (SMO). We tested the potential for molecular, cellular, and functional recovery in mice from the severe disruption of taste-organ biology and taste sensation that follows HH/SMO signaling inhibition. Sonidegib treatment led to rapid loss of taste buds (TB) in both fungiform and circumvallate papillae, including disruption of TB progenitor-cell proliferation and differentiation. Effects were selective, sparing nontaste papillae. To confirm that taste-organ effects of sonidegib treatment result from HH/SMO signaling inhibition, we studied mice with conditional global or epithelium-specific Smo deletions and observed similar effects. During sonidegib treatment, chorda tympani nerve responses to lingual chemical stimulation were maintained at 10 d but were eliminated after 16 d, associated with nearly complete TB loss. Notably, responses to tactile or cold stimulus modalities were retained. Further, innervation, which was maintained in the papilla core throughout treatment, was not sufficient to sustain TB during HH/SMO inhibition. Importantly, treatment cessation led to rapid and complete restoration of taste responses within 14 d associated with morphologic recovery in about 55% of TB. However, although taste nerve responses were sustained, TB were not restored in all fungiform papillae even with prolonged recovery for several months. This study establishes a physiologic, selective requirement for HH/SMO signaling in taste homeostasis that includes potential for sensory restoration and can explain the temporal recovery after taste dysgeusia in patients treated with HH/SMO inhibitors.
Keywords: chorda tympani nerve; circumvallate papilla; fungiform papilla; taste receptor cell; taste regeneration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Migden MR, et al. Treatment with two different doses of sonidegib in patients with locally advanced or metastatic basal cell carcinoma (BOLT): A multicentre, randomised, double-blind phase 2 trial. Lancet Oncol. 2015;16:716–728. - PubMed
-
- Rodon J, et al. A phase I, multicenter, open-label, first-in-human, dose-escalation study of the oral smoothened inhibitor Sonidegib (LDE225) in patients with advanced solid tumors. Clin Cancer Res. 2014;20:1900–1909. - PubMed
-
- Fife K, et al. Managing adverse events associated with vismodegib in the treatment of basal cell carcinoma. Future Oncol. 2017;13:175–184. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

