Endothelial TNF-α induction by Hsp60 secreted from THP-1 monocytes exposed to hyperglycaemic conditions
- PMID: 29134442
- PMCID: PMC6045554
- DOI: 10.1007/s12192-017-0858-x
Endothelial TNF-α induction by Hsp60 secreted from THP-1 monocytes exposed to hyperglycaemic conditions
Abstract
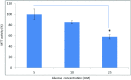
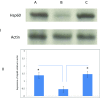
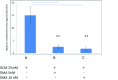
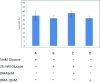
A non-resolving inflammation of the endothelium is recognised to be an important process leading to atherosclerosis. In diabetes, this process is thought to account for a significant number of cardiovascular disease-associated death and disability. However, the molecular mechanisms by which diabetes contributes to endothelial inflammation remain to be established. Whilst there is some evidence linking hyperglycaemia-induced reactive oxygen species (ROS) formation by the mitochondrial electron-transport chain to oxidative stress, cellular injury and apoptosis in the endothelium, a clear link to endothelium inflammation has not yet been established. The mitochondrial molecular stress protein Hsp60 is known to be secreted from mammalian cells and is capable of activating pro-inflammatory mediators on target cells expressing Toll-like receptors (TLRs). Hsp60 is also known to be elevated in serum of diabetes patients and has been shown to be upregulated by hyperglycaemic growth conditions in cultured human HeLa cells. This study shows that Hsp60 induced in human acute monocyte leukaemia cell line (THP-1) cells grown under hyperglycaemic conditions (25 mM glucose) was able to be secreted into growth media. Furthermore, the secretion of Hsp60 from THP-1 cells was able to be inhibited by 5,5-(N-N-dimethyl)-amiloride hydrochloride (DMA), an exosomal inhibitor. Interestingly, the conditioned media obtained from THP-1 cells grown in the presence of 25 mM glucose was able to induce the secretion of TNF-α in human vascular endothelium cell line (HUVEC). When conditioned media was immuno-depleted of Hsp60, there was a significant reduction in the release of TNF-α from the HUVEC cells. This suggests that a potential link may exist between hyperglycaemia-induced expression of Hsp60 in monocyte cells and vascular inflammation. Circulating levels of Hsp60 due to mitochondrial stress in diabetes patients could therefore be an important modulator of inflammation in endothelial cells and thus contribute to the increased incidences of atherosclerosis in diabetes mellitus.
Keywords: Endothelial cells; Hsp60; Hyperglycaemia; Inflammation; THP-1 monocytes; TNF-α.
Figures
References
-
- Asea AAA, De Maio A (2007) Heat shock proteins: potent mediators of inflammation and immunity: potent mediators of inflammation and immunity. Springer
-
- Campanella C, D'Anneo A, Gammazza AM, Bavisotto CC, Barone R, Emanuele S, Lo Cascio F, Mocciaro E, Fais S, De Macario EC, et al. The histone deacetylase inhibitor SAHA induces HSP60 nitration and its extracellular release by exosomal vesicles in human lung-derived carcinoma cells. Oncotarget. 2015;7:28849–28867. - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

