High-grade cervical intraepithelial neoplasia in human papillomavirus self-sampling of screening non-attenders
- PMID: 29136403
- PMCID: PMC5765223
- DOI: 10.1038/bjc.2017.371
High-grade cervical intraepithelial neoplasia in human papillomavirus self-sampling of screening non-attenders
Abstract
Background: Self-sampling for human papillomavirus (HPV) offered to women who do not participate in cervical cancer screening is an increasingly popular method to increase screening coverage. The rationale behind self-sampling is that unscreened women harbour a high proportion of undetected precancer lesions. Here, we compare the cervical intraepithelial neoplasia grade 2 or worse (⩾CIN2) detection rate between non-attenders who participated in self-sampling and women attending routine screening.
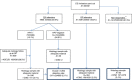
Methods: A total of 23 632 women who were qualified as non-attenders in the Copenhagen Region were invited for HPV-based self-sampling. Of these, 4824 women returned a self-sample, and HPV-positive women were referred for cytology and HPV co-testing as follow-up. The entire cohort and a reference cohort (3347 routinely screened women) were followed for histopathology confirmed ⩾CIN2. Odds ratio (OR) and the relative positive predictive value of ⩾CIN2 detection between the two populations were estimated.
Results: Women participating in self-sampling had a higher ⩾CIN2 detection than women undergoing routine cytology-based screening (OR=1.83, 95% CI: 1.21-2.77) and a similar detection as routinely screened women tested with cytology and HPV testing (OR=1.03, 95% CI: 0.75-1.40). The positive predictive value for ⩾CIN2 was higher in screening non-attenders than in routinely HPV- and cytology-screened screened women (36.5% vs 25.6%, respectively).
Conclusions: Self-sampling offered to non-attenders showed higher detection rates for ⩾CIN2 than routine cytology-based screening, and similar detection rates as HPV and cytology co-testing. This reinforces the importance of self-sampling for screening non-attenders in organised cervical cancer screening.
Conflict of interest statement
JUHL attended meetings with various HPV device manufacturers. MR attended meetings with various HPV device manufacturers. She and her former employer received fees for lectures on her behalf from Qiagen. DME attended meetings with various HPV device manufacturers. She received honoraria from BD and Qiagen for lectures. HP attended meetings with various HPV device manufacturers. CR participated in meetings with Roche with fee paid to the University of Copenhagen. EL participated in meetings with Roche and Astra-Zeneca with fees paid to the University of Copenhagen, and was unpaid advisor to GenProbe and NorChip. Roche has provided test kits to Trial23. SKK has received lecture fees, scientific advisory board fees from Merck, Sanofi Pasteur MSD, BD, and unrestricted research grants through the affiliating institute from Merck. JD has received grants to his institution for studies on HPV vaccination from Merck/SPMSD. JB attended meetings with various HPV device manufacturers. He used to serve as a paid advisor to Roche and Genomica, and has received honoraria from Hologic/Gen-Probe, Roche, Qiagen, Genomica, and BD Diagnostics for lectures. He is principal investigator on studies funded by BD diagnostics. Hvidovre Hospital holds a recompense agreement with Genomica on a KRAS/BRAF diagnostic system. KME and KEJ declare no conflict of interest.
Figures

References
-
- Bais AG, van Kemenade FJ, Berkhof J, Verheijen RH, Snijders PJ, Voorhorst F, Babovic M, van Ballegooijen M, Helmerhorst TJ, Meijer CJ (2007) Human papillomavirus testing on self-sampled cervicovaginal brushes: an effective alternative to protect nonresponders in cervical screening programs. Int J Cancer 120(7): 1505–1510. - PubMed
-
- Bjerregaard B, Larsen OB (2011) The Danish Pathology Register. Scand J Public Health 39(7 Suppl): 72–74. - PubMed
-
- (2014) Danish Quality Assurance Database for the Cervical Cancer Screening Program Annual report 2014 Vol. 2015.
-
- Dugue PA, Lynge E, Bjerregaard B, Rebolj M (2012) Non-participation in screening: the case of cervical cancer in Denmark. Prev Med 54(3-4): 266–269. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

