In vivo bioluminescence imaging of labile iron accumulation in a murine model of Acinetobacter baumannii infection
- PMID: 29138321
- PMCID: PMC5715752
- DOI: 10.1073/pnas.1708747114
In vivo bioluminescence imaging of labile iron accumulation in a murine model of Acinetobacter baumannii infection
Abstract
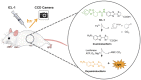
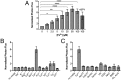
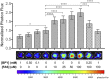
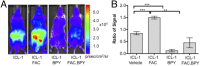
Iron is an essential metal for all organisms, yet disruption of its homeostasis, particularly in labile forms that can contribute to oxidative stress, is connected to diseases ranging from infection to cancer to neurodegeneration. Iron deficiency is also among the most common nutritional deficiencies worldwide. To advance studies of iron in healthy and disease states, we now report the synthesis and characterization of iron-caged luciferin-1 (ICL-1), a bioluminescent probe that enables longitudinal monitoring of labile iron pools (LIPs) in living animals. ICL-1 utilizes a bioinspired endoperoxide trigger to release d-aminoluciferin for selective reactivity-based detection of Fe2+ with metal and oxidation state specificity. The probe can detect physiological changes in labile Fe2+ levels in live cells and mice experiencing iron deficiency or overload. Application of ICL-1 in a model of systemic bacterial infection reveals increased iron accumulation in infected tissues that accompany transcriptional changes consistent with elevations in both iron acquisition and retention. The ability to assess iron status in living animals provides a powerful technology for studying the contributions of iron metabolism to physiology and pathology.
Keywords: infectious disease; labile iron; luciferin; metal homeostasis; molecular imaging.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Cammack R, Wrigglesworth JM, Baum H. Transport and Storage. CRC; Boca Raton, FL: 1989.
-
- Andrews NC. Iron homeostasis: Insights from genetics and animal models. Nat Rev Genet. 2000;1:208–217. - PubMed
-
- Winterbourn CC. Toxicity of iron and hydrogen peroxide: The Fenton reaction. Toxicol Lett. 1995;82-83:969–974. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

