Carbon source-sink relationship in Arabidopsis thaliana: the role of sucrose transporters
- PMID: 29138971
- PMCID: PMC5809531
- DOI: 10.1007/s00425-017-2807-4
Carbon source-sink relationship in Arabidopsis thaliana: the role of sucrose transporters
Abstract
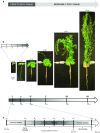
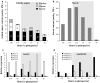
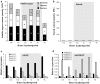
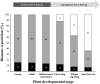
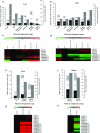
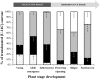
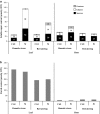
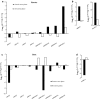
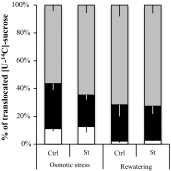
The regulation of source-to-sink sucrose transport is associated with AtSUC and AtSWEET sucrose transporters' gene expression changes in plants grown hydroponically under different physiological conditions. Source-to-sink transport of sucrose is one of the major determinants of plant growth. Whole-plant carbohydrates' partitioning requires the specific activity of membrane sugar transporters. In Arabidopsis thaliana plants, two families of transporters are involved in sucrose transport: AtSUCs and AtSWEETs. This study is focused on the comparison of sucrose transporter gene expression, soluble sugar and starch levels and long distance sucrose transport, in leaves and sink organs (mainly roots) in different physiological conditions (along the plant life cycle, during a diel cycle, and during an osmotic stress) in plants grown hydroponically. In leaves, the AtSUC2, AtSWEET11, and 12 genes known to be involved in phloem loading were highly expressed when sucrose export was high and reduced during osmotic stress. In roots, AtSUC1 was highly expressed and its expression profile in the different conditions tested suggests that it may play a role in sucrose unloading in roots and in root growth. The SWEET transporter genes AtSWEET12, 13, and 15 were found expressed in all organs at all stages studied, while differential expression was noticed for AtSWEET14 in roots, stems, and siliques and AtSWEET9, 10 expressions were only detected in stems and siliques. A role for these transporters in carbohydrate partitioning in different source-sink status is proposed, with a specific attention on carbon demand in roots. During development, despite trophic competition with others sinks, roots remained a significant sink, but during osmotic stress, the amount of translocated [U-14C]-sucrose decreased for rosettes and roots. Altogether, these results suggest that source-sink relationship may be linked with the regulation of sucrose transporter gene expression.
Keywords: Diel cycle; Full life development; Hydroponic culture; Osmotic stress; Roots; Sugar transporter gene expression.
Figures

References
-
- Ali ML, Luetchens J, Singh A, Shaver T, Kruger G, Lorenz A. Greenhouse screening of maize genotypes for deep root mass and related root traits and their association with grain yield under water-deficit conditions in the field. Euphytica. 2016;207(1):79–94. doi: 10.1007/s10681-015-1533-x. - DOI
-
- Angeles-Núñez JG, Tiessen A. Mutation of the transcription factor LEAFY COTYLEDON 2 alters the chemical composition of Arabidopsis seeds, decreasing oil and protein content, while maintaining high levels of starch and sucrose in mature seeds. J Plant Physiol. 2011;168(16):1891–1900. doi: 10.1016/j.jplph.2011.05.003. - DOI - PubMed
-
- Arteca RN, Arteca JM. A novel method for growing Arabidopsis thaliana plants hydroponically. Physiol Plant. 2000;108(2):188–193. doi: 10.1034/j.1399-3054.2000.108002188.x. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

