Potent and selective inhibition of SH3 domains with dirhodium metalloinhibitors
- PMID: 29142714
- PMCID: PMC5667506
- DOI: 10.1039/c5sc01602a
Potent and selective inhibition of SH3 domains with dirhodium metalloinhibitors
Abstract
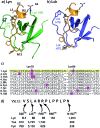
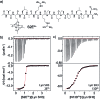
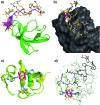
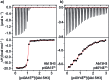
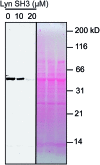
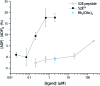
Src-family kinases (SFKs) play important roles in human biology and are key drug targets as well. However, achieving selective inhibition of individual Src-family kinases is challenging due to the high similarity within the protein family. We describe rhodium(ii) conjugates that deliver both potent and selective inhibition of Src-family SH3 domains. Rhodium(ii) conjugates offer dramatic affinity enhancements due to interactions with specific and unique Lewis-basic histidine residues near the SH3 binding interface, allowing predictable, structure-guided inhibition of SH3 targets that are recalcitrant to traditional inhibitors. In one example, a simple metallopeptide binds the Lyn SH3 domain with 6 nM affinity and exhibits functional activation of Lyn kinase under biologically relevant concentrations (EC50 ∼ 200 nM).
Figures
Similar articles
-
Aminoquinoline-Rhodium(II) Conjugates as Src-Family SH3 Ligands.ACS Med Chem Lett. 2019 Sep 9;10(10):1380-1385. doi: 10.1021/acsmedchemlett.9b00309. eCollection 2019 Oct 10. ACS Med Chem Lett. 2019. PMID: 31620222 Free PMC article.
-
Differential sensitivity of Src-family kinases to activation by SH3 domain displacement.PLoS One. 2014 Aug 21;9(8):e105629. doi: 10.1371/journal.pone.0105629. eCollection 2014. PLoS One. 2014. PMID: 25144189 Free PMC article.
-
The Two Isoforms of Lyn Display Different Intramolecular Fuzzy Complexes with the SH3 Domain.Molecules. 2018 Oct 23;23(11):2731. doi: 10.3390/molecules23112731. Molecules. 2018. PMID: 30360468 Free PMC article.
-
Recent progress of Src SH2 and SH3 inhibitors as anticancer agents.Curr Med Chem. 2010;17(12):1117-24. doi: 10.2174/092986710790827861. Curr Med Chem. 2010. PMID: 20158477 Review.
-
Src family kinases: regulation of their activities, levels and identification of new pathways.Biochim Biophys Acta. 2008 Jan;1784(1):56-65. doi: 10.1016/j.bbapap.2007.08.012. Epub 2007 Aug 22. Biochim Biophys Acta. 2008. PMID: 17905674 Review.
Cited by
-
Directed Evolution of a Highly Specific FN3 Monobody to the SH3 Domain of Human Lyn Tyrosine Kinase.PLoS One. 2016 Jan 5;11(1):e0145872. doi: 10.1371/journal.pone.0145872. eCollection 2016. PLoS One. 2016. PMID: 26731115 Free PMC article.
-
Targeting STAT3 anti-apoptosis pathways with organic and hybrid organic-inorganic inhibitors.Org Biomol Chem. 2020 May 6;18(17):3288-3296. doi: 10.1039/c9ob02682g. Org Biomol Chem. 2020. PMID: 32286579 Free PMC article.
-
Crystal structure of the SH3 domain of human Lyn non-receptor tyrosine kinase.PLoS One. 2019 Apr 10;14(4):e0215140. doi: 10.1371/journal.pone.0215140. eCollection 2019. PLoS One. 2019. PMID: 30969999 Free PMC article.
-
Aminoquinoline-Rhodium(II) Conjugates as Src-Family SH3 Ligands.ACS Med Chem Lett. 2019 Sep 9;10(10):1380-1385. doi: 10.1021/acsmedchemlett.9b00309. eCollection 2019 Oct 10. ACS Med Chem Lett. 2019. PMID: 31620222 Free PMC article.
-
New Heteroleptic Ruthenium(II) Complexes with Sulfamethoxypyridazine and Diimines as Potential Antitumor Agents.Molecules. 2019 Jun 7;24(11):2154. doi: 10.3390/molecules24112154. Molecules. 2019. PMID: 31181667 Free PMC article.
References
-
- Schreiner S. J., Schiavone A. P., Smithgall T. E. J. Biol. Chem. 2002;277:45680–45687. - PubMed
-
- Ingley E., Sarna M. K., Beaumont J. G., Tilbrook P. A., Tsai S., Takemoto Y., Williams J. H., Klinken S. P. J. Biol. Chem. 2000;275:7887–7893. - PubMed
-
- Takemoto Y., Sato M., Furuta M., Hashimoto Y. Int. Immunol. 1996;8:1699–1705. - PubMed
-
- Moarefi I., LaFevre-Bernt M., Sicheri F., Huse M., Lee C.-H., Kuriyan J., Miller W. T. Nature. 1997;385:650–653. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

