Buserelin alleviates chloride transport defect in human cystic fibrosis nasal epithelial cells
- PMID: 29145426
- PMCID: PMC5690610
- DOI: 10.1371/journal.pone.0187774
Buserelin alleviates chloride transport defect in human cystic fibrosis nasal epithelial cells
Abstract
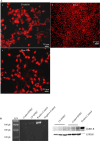
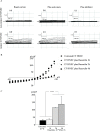
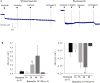
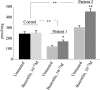
Cystic fibrosis (CF) is the most common autosomal recessive disease in Caucasians caused by mutations in the gene encoding the Cystic Fibrosis Transmembrane conductance Regulator (CFTR) chloride (Cl-) channel regulated by protein kinases, phosphatases, divalent cations and by protein-protein interactions. Among protein-protein interactions, we previously showed that Annexin A5 (AnxA5) binds to CFTR and is involved in the channel localization within membranes and in its Cl- channel function. The deletion of phenylalanine at position 508 (F508del) is the most common mutation in CF which leads to an altered protein (F508del-CFTR) folding with a nascent protein retained within the ER and is quickly degraded. We previously showed that AnxA5 binds to F508del-CFTR and that its increased expression due to a Gonadoliberin (GnRH) augments Cl- efflux in cells expressing F508del-CFTR. The aim of the present work was to use the GnRH analog buserelin which is already used in medicine. Human nasal epithelial cells from controls and CF patients (F508del/F508del) were treated with buserelin and we show here that the treatment alleviates Cl- channel defects in CF cells. Using proteomics we highlighted some proteins explaining this result. Finally, we propose that buserelin is a potential new pharmaceutical compound that can be used in CF and that bronchus can be targeted since we show here that they express GnRH-R.
Conflict of interest statement
Figures


Similar articles
-
Improvement of chloride transport defect by gonadotropin-releasing hormone (GnRH) in cystic fibrosis epithelial cells.PLoS One. 2014 Feb 19;9(2):e88964. doi: 10.1371/journal.pone.0088964. eCollection 2014. PLoS One. 2014. PMID: 24586461 Free PMC article.
-
F508del-CFTR increases intracellular Ca(2+) signaling that causes enhanced calcium-dependent Cl(-) conductance in cystic fibrosis.Biochim Biophys Acta. 2011 Nov;1812(11):1385-92. doi: 10.1016/j.bbadis.2011.08.008. Epub 2011 Aug 30. Biochim Biophys Acta. 2011. PMID: 21907281
-
Correction of chloride transport and mislocalization of CFTR protein by vardenafil in the gastrointestinal tract of cystic fibrosis mice.PLoS One. 2013 Oct 24;8(10):e77314. doi: 10.1371/journal.pone.0077314. eCollection 2013. PLoS One. 2013. PMID: 24204804 Free PMC article.
-
The cystic fibrosis transmembrane conductance regulator (CFTR) and its stability.Cell Mol Life Sci. 2017 Jan;74(1):23-38. doi: 10.1007/s00018-016-2386-8. Epub 2016 Oct 12. Cell Mol Life Sci. 2017. PMID: 27734094 Free PMC article. Review.
-
Targeting F508del-CFTR to develop rational new therapies for cystic fibrosis.Acta Pharmacol Sin. 2011 Jun;32(6):693-701. doi: 10.1038/aps.2011.71. Acta Pharmacol Sin. 2011. PMID: 21642944 Free PMC article. Review.
Cited by
-
A novel role for CFTR interaction with LH and FGF in azoospermia and epididymal maldevelopment caused by cryptorchidism.Basic Clin Androl. 2022 Jun 21;32(1):10. doi: 10.1186/s12610-022-00160-0. Basic Clin Androl. 2022. PMID: 35725394 Free PMC article. Review.
References
-
- Riordan JR, Rommens JM, Kerem B, Alon N, Rozmahel R, Grzelczak Z et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science (1989); 245:1066–1073. - PubMed
-
- Bear CE, Li CH, Kartner N, Bridges RJ, Jensen TJ, Ramjeesingh M et al. Purification and functional reconstitution of the cystic fibrosis transmembrane conductance regulator (CFTR). Cell (1992); 68:809–818. - PubMed
-
- Hwang TC, Sheppard DN. Gating of the CFTR Cl− channel by ATP-driven nucleotide-binding domain dimerisation. J Physiol. (2009); 587:2151–2161. doi: 10.1113/jphysiol.2009.171595 - DOI - PMC - PubMed
-
- Ostedgaard LS, Rogers CS, Dong Q, Randak CO, Vermeer DW, Rokhlina T et al. Processing and function of CFTR-ΔF508 are species-dependent. Proc Natl Acad Sci USA (2007); 104:15370–15375. doi: 10.1073/pnas.0706974104 - DOI - PMC - PubMed
-
- Miki H, Zhou Z, Li M, Hwang TC, Bompadre SG. Potentiation of disease-associated cystic fibrosis transmembrane conductance regulator mutants by hydrolyzable ATP analogs. J Biol Chem. (2010); 285:19967–19975. doi: 10.1074/jbc.M109.092684 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

