The somatically generated portion of T cell receptor CDR3α contributes to the MHC allele specificity of the T cell receptor
- PMID: 29148973
- PMCID: PMC5701794
- DOI: 10.7554/eLife.30918
The somatically generated portion of T cell receptor CDR3α contributes to the MHC allele specificity of the T cell receptor
Abstract
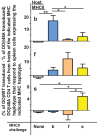
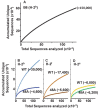
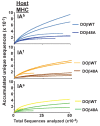
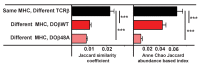
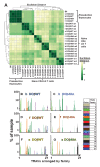
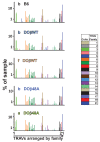
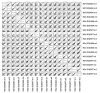
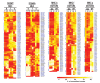
Mature T cells bearing αβ T cell receptors react with foreign antigens bound to alleles of major histocompatibility complex proteins (MHC) that they were exposed to during their development in the thymus, a phenomenon known as positive selection. The structural basis for positive selection has long been debated. Here, using mice expressing one of two different T cell receptor β chains and various MHC alleles, we show that positive selection-induced MHC bias of T cell receptors is affected both by the germline encoded elements of the T cell receptor α and β chain and, surprisingly, dramatically affected by the non germ line encoded portions of CDR3 of the T cell receptor α chain. Thus, in addition to determining specificity for antigen, the non germline encoded elements of T cell receptors may help the proteins cope with the extremely polymorphic nature of major histocompatibility complex products within the species.
Keywords: CDR3 alpha; T cell receptor; immunology; major histocompatibility complex; mouse; selection; thymus.
Conflict of interest statement
No competing interests declared.
Figures


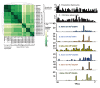

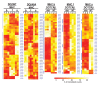

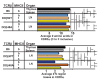
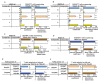


References
-
- Adams JJ, Narayanan S, Birnbaum ME, Sidhu SS, Blevins SJ, Gee MH, Sibener LV, Baker BM, Kranz DM, Garcia KC. Structural interplay between germline interactions and adaptive recognition determines the bandwidth of TCR-peptide-MHC cross-reactivity. Nature Immunology. 2016;17:87–94. doi: 10.1038/ni.3310. - DOI - PMC - PubMed
-
- Allison JP, McIntyre BW, Bloch D. Tumor-specific antigen of murine T-lymphoma defined with monoclonal antibody. Journal of Immunology. 1982;129:2293–2300. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

