Mutations in RAB39B in individuals with intellectual disability, autism spectrum disorder, and macrocephaly
- PMID: 29152164
- PMCID: PMC5679329
- DOI: 10.1186/s13229-017-0175-3
Mutations in RAB39B in individuals with intellectual disability, autism spectrum disorder, and macrocephaly
Abstract
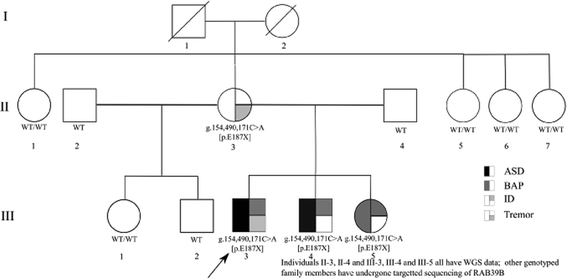
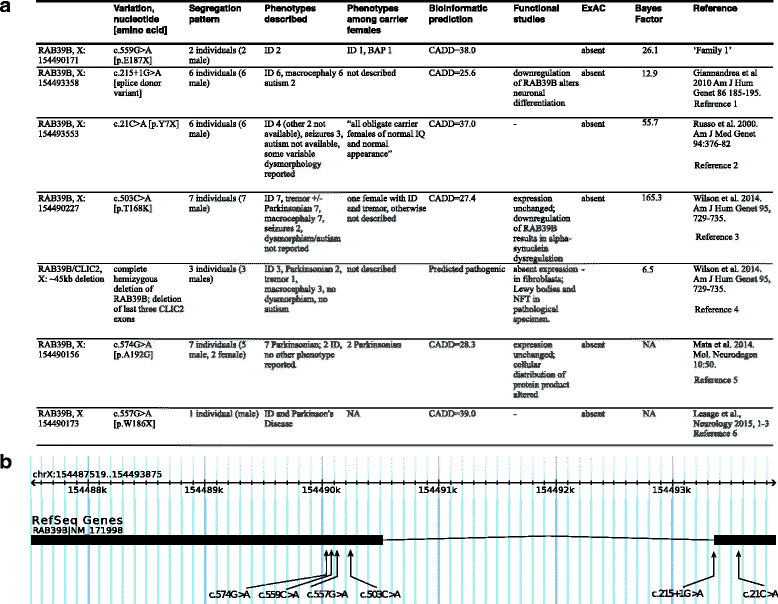
Background: Autism spectrum disorder (ASD), a developmental disorder of early childhood onset, affects males four times more frequently than females, suggesting a role for the sex chromosomes. In this study, we describe a family with ASD in which a predicted pathogenic nonsense mutation in the X-chromosome gene RAB39B segregates with ASD phenotype.
Methods: Clinical phenotyping, microarray, and whole genome sequencing (WGS) were performed on the five members of this family. Maternal and female sibling X inactivation ratio was calculated, and phase was investigated. Mutant-induced pluripotent stem cells engineered for an exon 2 nonsense mutation were generated and differentiated into cortical neurons for expression and pathway analyses.
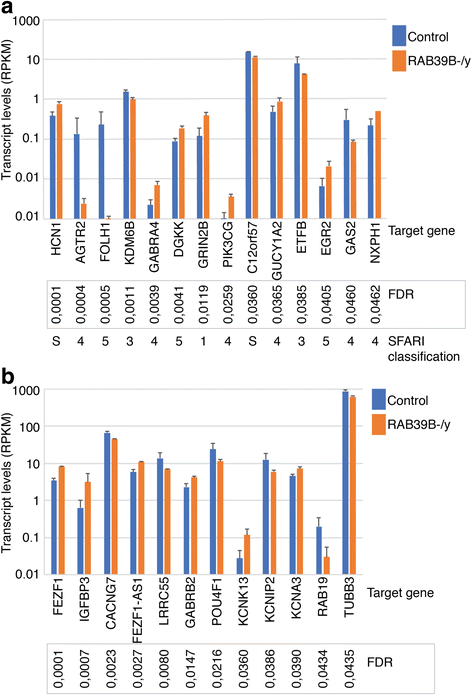
Results: Two males with an inherited RAB39B mutation both presented with macrocephaly, intellectual disability (ID), and ASD. Their female sibling with the same mutation presented with ID and a broad autism phenotype. In contrast, their transmitting mother has no neurodevelopmental diagnosis. Our investigation of phase indicated maternal preferential inactivation of the mutated allele, with no such bias observed in the female sibling. We offer the explanation that this bias in X inactivation may explain the absence of a neurocognitive phenotype in the mother. Our cellular knockout model of RAB39B revealed an impact on expression in differentiated neurons for several genes implicated in brain development and function, supported by our pathway enrichment analysis.
Conclusions: Penetrance for ASD is high among males but more variable among females with RAB39B mutations. A critical role for this gene in brain development and function is demonstrated.
Keywords: Intellectual disability (ID); RAB39B; RNAseq; Whole genome sequencing (WGS).
Conflict of interest statement
Ethics approval and consent to participate
All data were collected following informed consent from participants or substitute decision-makers, and the study was conducted with approval from the Hospital for Sick Children, Toronto, local research ethics board.
Consent for publication
The mother has provided specific written consent for this case report using the Hospital for Sick Children consent form.
Competing interests
The authors declare no conflict of interest for the contents of the data in this study.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Cerebral organoid and mouse models reveal a RAB39b-PI3K-mTOR pathway-dependent dysregulation of cortical development leading to macrocephaly/autism phenotypes.Genes Dev. 2020 Apr 1;34(7-8):580-597. doi: 10.1101/gad.332494.119. Epub 2020 Feb 27. Genes Dev. 2020. PMID: 32115408 Free PMC article.
-
De novo nonsense and frameshift variants of TCF20 in individuals with intellectual disability and postnatal overgrowth.Eur J Hum Genet. 2016 Dec;24(12):1739-1745. doi: 10.1038/ejhg.2016.90. Epub 2016 Jul 20. Eur J Hum Genet. 2016. PMID: 27436265 Free PMC article.
-
Clinical-Genetic Approach to Conditions with Macrocephaly and ASD/Behaviour Abnormalities: Variants in PTEN and PPP2R5D Are the Most Recurrent Gene Mutations in a Patient-Oriented Diagnostic Strategy.Genes (Basel). 2025 Apr 20;16(4):469. doi: 10.3390/genes16040469. Genes (Basel). 2025. PMID: 40282429 Free PMC article.
-
RAB39B's role in membrane traffic, autophagy, and associated neuropathology.J Cell Physiol. 2021 Mar;236(3):1579-1592. doi: 10.1002/jcp.29962. Epub 2020 Aug 6. J Cell Physiol. 2021. PMID: 32761840 Review.
-
A novel HIST1HE pathogenic variant in a girl with macrocephaly and intellectual disability: a new case and review of literature.Clin Dysmorphol. 2021 Jan;30(1):39-43. doi: 10.1097/MCD.0000000000000352. Clin Dysmorphol. 2021. PMID: 33086257 Review.
Cited by
-
Exome-wide analysis identify multiple variations in olfactory receptor genes (OR12D2 and OR5V1) associated with autism spectrum disorder in Saudi females.Front Med (Lausanne). 2023 Feb 1;10:1051039. doi: 10.3389/fmed.2023.1051039. eCollection 2023. Front Med (Lausanne). 2023. PMID: 36817779 Free PMC article.
-
Cerebral organoid and mouse models reveal a RAB39b-PI3K-mTOR pathway-dependent dysregulation of cortical development leading to macrocephaly/autism phenotypes.Genes Dev. 2020 Apr 1;34(7-8):580-597. doi: 10.1101/gad.332494.119. Epub 2020 Feb 27. Genes Dev. 2020. PMID: 32115408 Free PMC article.
-
shinyseg: a web application for flexible cosegregation and sensitivity analysis.Bioinformatics. 2024 May 2;40(5):btae201. doi: 10.1093/bioinformatics/btae201. Bioinformatics. 2024. PMID: 38598476 Free PMC article.
-
Cntnap2-dependent molecular networks in autism spectrum disorder revealed through an integrative multi-omics analysis.Mol Psychiatry. 2023 Feb;28(2):810-821. doi: 10.1038/s41380-022-01822-1. Epub 2022 Oct 17. Mol Psychiatry. 2023. PMID: 36253443 Free PMC article.
-
Detection of copy number variants and genes by chromosomal microarray in an Emirati neurodevelopmental disorders cohort.Neurogenetics. 2022 Apr;23(2):137-149. doi: 10.1007/s10048-022-00689-2. Epub 2022 Mar 24. Neurogenetics. 2022. PMID: 35325322
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

