Nickel-catalyzed transamidation of aliphatic amide derivatives
- PMID: 29163929
- PMCID: PMC5632791
- DOI: 10.1039/c7sc01980g
Nickel-catalyzed transamidation of aliphatic amide derivatives
Abstract
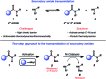
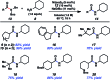
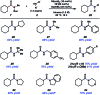
Transamidation, or the conversion of one amide to another, is a long-standing challenge in organic synthesis. Although notable progress has been made in the transamidation of primary amides, the transamidation of secondary amides has remained underdeveloped, especially when considering aliphatic substrates. Herein, we report a two-step approach to achieve the transamidation of secondary aliphatic amides, which relies on non-precious metal catalysis. The method involves initial Boc-functionalization of secondary amide substrates to weaken the amide C-N bond. Subsequent treatment with a nickel catalyst, in the presence of an appropriate amine coupling partner, then delivers the net transamidated products. The transformation proceeds in synthetically useful yields across a range of substrates. A series of competition experiments delineate selectivity patterns that should influence future synthetic design. Moreover, the transamidation of Boc-activated secondary amide derivatives bearing epimerizable stereocenters underscores the mildness and synthetic utility of this methodology. This study provides the most general solution to the classic problem of secondary amide transamidation reported to date.
Figures



Similar articles
-
A two-step approach to achieve secondary amide transamidation enabled by nickel catalysis.Nat Commun. 2016 May 20;7:11554. doi: 10.1038/ncomms11554. Nat Commun. 2016. PMID: 27199089 Free PMC article.
-
Nickel-Catalyzed Suzuki-Miyaura Coupling of Aliphatic Amides.ACS Catal. 2018 Feb 2;8(2):1003-1008. doi: 10.1021/acscatal.7b03688. Epub 2017 Dec 20. ACS Catal. 2018. PMID: 29682398 Free PMC article.
-
Nickel-Catalyzed Esterification of Aliphatic Amides.Angew Chem Int Ed Engl. 2016 Nov 21;55(48):15129-15132. doi: 10.1002/anie.201607856. Epub 2016 Nov 4. Angew Chem Int Ed Engl. 2016. PMID: 27813308 Free PMC article.
-
Direct Transamidation Reactions: Mechanism and Recent Advances.Molecules. 2018 Sep 18;23(9):2382. doi: 10.3390/molecules23092382. Molecules. 2018. PMID: 30231486 Free PMC article. Review.
-
Transition-Metal-Free Activation of Amides by N-C Bond Cleavage.Chem Rec. 2020 Jul;20(7):649-659. doi: 10.1002/tcr.201900072. Epub 2019 Dec 13. Chem Rec. 2020. PMID: 31833633 Review.
Cited by
-
Breaking Amide C-N Bonds in an Undergraduate Organic Chemistry Laboratory.J Chem Educ. 2019 Apr 9;96(4):776-780. doi: 10.1021/acs.jchemed.8b00489. Epub 2019 Mar 6. J Chem Educ. 2019. PMID: 31073247 Free PMC article.
-
Efficient cleavage of tertiary amide bonds via radical-polar crossover using a copper(ii) bromide/Selectfluor hybrid system.Chem Sci. 2020 Oct 14;11(45):12323-12328. doi: 10.1039/d0sc05137c. Chem Sci. 2020. PMID: 34094440 Free PMC article.
-
Ni-Catalyzed Suzuki-Miyaura Cross-Coupling of Aliphatic Amides on the Benchtop.Org Lett. 2020 Jan 3;22(1):1-5. doi: 10.1021/acs.orglett.9b03434. Epub 2019 Oct 17. Org Lett. 2020. PMID: 31621338 Free PMC article.
-
Highly selective transition-metal-free transamidation of amides and amidation of esters at room temperature.Nat Commun. 2018 Oct 9;9(1):4165. doi: 10.1038/s41467-018-06623-1. Nat Commun. 2018. PMID: 30302003 Free PMC article.
-
Amide N-C Bond Activation: A Graphical Overview of Acyl and Decarbonylative Coupling.SynOpen. 2023 Feb;7(1):88-101. doi: 10.1055/a-2035-6733. Epub 2023 Mar 6. SynOpen. 2023. PMID: 38037650 Free PMC article.
References
-
- For a recent review on amide bond formation, see: Figueiredo R. M., Suppo J.-S., Campagne J.-M., Chem. Rev., 2016, 116 , 12029 –12122 . - PubMed
-
-
For primary amide transamidations, see:
- Dineen T. A., Zajac M. A., Myers A. G. J. Am. Chem. Soc. 2006;128:1419–1422. - PubMed
- Allen C. L., Atkinson B. N., Williams J. M. J. Angew. Chem., Int. Ed. 2012;51:1383–1386. - PubMed
- Becerra-Figueroa L., Ojeda-Porras A., Gamba-Sánchez D. J. Org. Chem. 2014;79:4544–4552. - PubMed
- Rao S. N., Mohan D. C., Adimurthy S. Green Chem. 2014;16:4122–4126.
- Rao S. N., Mohan D. C., Adimurthy S. Org. Lett. 2013;15:1496–1499. - PubMed
- Rao S. N., Mohan D. C., Adimurthy S. RSC Adv. 2015;5:95313–95317.
-
-
- Mucsi Z., Chass G. A., Csizmadia I. G. J. Phys. Chem. B. 2008;112:7885–7893. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

