Blood-borne interleukin-1β acts on the subfornical organ to upregulate the sympathoexcitatory milieu of the hypothalamic paraventricular nucleus
- PMID: 29167166
- PMCID: PMC5899248
- DOI: 10.1152/ajpregu.00211.2017
Blood-borne interleukin-1β acts on the subfornical organ to upregulate the sympathoexcitatory milieu of the hypothalamic paraventricular nucleus
Abstract
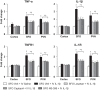
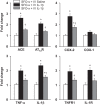
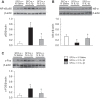
We previously reported that microinjection of the proinflammatory cytokine interleukin-1β (IL-1β) into the subfornical organ (SFO) elicits a pressor response accompanied by increases in inflammation and renin-angiotensin system (RAS) activity in the SFO and hypothalamic paraventricular nucleus (PVN). The present study sought to determine whether blood-borne IL-1β induces similar neurochemical changes in the SFO and PVN and, if so, whether increased inflammation and RAS activity at the SFO level orchestrate the sympathoexcitatory response to circulating IL-1β. In urethane-anesthetized male Sprague-Dawley rats, intravenous injection of IL-1β (500 ng) increased blood pressure, heart rate, renal sympathetic nerve activity, and mRNA for angiotensin-converting enzyme, angiotensin II type 1a receptor, cyclooxygenase-2, tumor necrosis factor-α, and IL-1β, as well as the tumor necrosis factor-α p55 receptor and the IL-1 receptor, in the SFO and PVN. Pretreatment with SFO microinjections of the angiotensin II type 1a receptor blocker losartan (1 µg), the angiotensin-converting enzyme inhibitor captopril (1 µg), or the cyclooxygenase-2 inhibitor NS-398 (2 µg) attenuated expression of these excitatory mediators in the SFO and downstream in the PVN and the IL-1β-induced pressor responses. An SFO lesion minimized the IL-1β-induced expression of inflammatory and RAS components as well as c-Fos, an indicator of neuronal excitation, in the PVN. These studies demonstrate that circulating IL-1β, which increases in cardiovascular disorders such as hypertension and heart failure, acts on the SFO to increase inflammation and RAS activity in the SFO and PVN and that intervening in these neurochemical processes in the SFO can significantly reduce the sympathetic response.
Keywords: brain; cyclooxygenase-2; proinflammatory cytokines; renin-angiotensin system; sympathetic nervous system.
Figures



References
-
- Bergman MR, Holycross BJ. Pharmacological modulation of myocardial tumor necrosis factor-α production by phosphodiesterase inhibitors. J Pharmacol Exp Ther 279: 247–254, 1996. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

