The first cell-fate decision of mouse preimplantation embryo development: integrating cell position and polarity
- PMID: 29167310
- PMCID: PMC5717349
- DOI: 10.1098/rsob.170210
The first cell-fate decision of mouse preimplantation embryo development: integrating cell position and polarity
Abstract
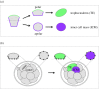
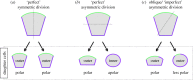
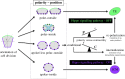
During the first cell-fate decision of mouse preimplantation embryo development, a population of outer-residing polar cells is segregated from a second population of inner apolar cells to form two distinct cell lineages: the trophectoderm and the inner cell mass (ICM), respectively. Historically, two models have been proposed to explain how the initial differences between these two cell populations originate and ultimately define them as the two stated early blastocyst stage cell lineages. The 'positional' model proposes that cells acquire distinct fates based on differences in their relative position within the developing embryo, while the 'polarity' model proposes that the differences driving the lineage segregation arise as a consequence of the differential inheritance of factors, which exhibit polarized subcellular localizations, upon asymmetric cell divisions. Although these two models have traditionally been considered separately, a growing body of evidence, collected over recent years, suggests the existence of a large degree of compatibility. Accordingly, the main aim of this review is to summarize the major historical and more contemporarily identified events that define the first cell-fate decision and to place them in the context of both the originally proposed positional and polarity models, thus highlighting their functional complementarity in describing distinct aspects of the developmental programme underpinning the first cell-fate decision in mouse embryogenesis.
Keywords: cell positioning and polarity; cell-fate; preimplantation mouse embryo.
© 2017 The Authors.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Revisiting trophectoderm-inner cell mass lineage segregation in the mammalian preimplantation embryo.Hum Reprod. 2024 Sep 1;39(9):1889-1898. doi: 10.1093/humrep/deae142. Hum Reprod. 2024. PMID: 38926157 Free PMC article. Review.
-
The first two cell-fate decisions of preimplantation mouse embryo development are not functionally independent.Sci Rep. 2015 Oct 13;5:15034. doi: 10.1038/srep15034. Sci Rep. 2015. PMID: 26461180 Free PMC article.
-
Cell Polarity-Dependent Regulation of Cell Allocation and the First Lineage Specification in the Preimplantation Mouse Embryo.Curr Top Dev Biol. 2018;128:11-35. doi: 10.1016/bs.ctdb.2017.10.008. Epub 2017 Nov 22. Curr Top Dev Biol. 2018. PMID: 29477161 Review.
-
Position- and polarity-dependent Hippo signaling regulates cell fates in preimplantation mouse embryos.Semin Cell Dev Biol. 2015 Dec;47-48:80-7. doi: 10.1016/j.semcdb.2015.05.003. Epub 2015 May 15. Semin Cell Dev Biol. 2015. PMID: 25986053 Review.
-
Mechanisms of trophectoderm fate specification in preimplantation mouse development.Dev Growth Differ. 2010 Apr;52(3):263-73. doi: 10.1111/j.1440-169X.2009.01158.x. Epub 2010 Jan 20. Dev Growth Differ. 2010. PMID: 20100249 Review.
Cited by
-
Horse ooplasm supports in vitro preimplantation development of zebra ICSI and SCNT embryos without compromising YAP1 and SOX2 expression pattern.PLoS One. 2020 Sep 11;15(9):e0238948. doi: 10.1371/journal.pone.0238948. eCollection 2020. PLoS One. 2020. PMID: 32915925 Free PMC article.
-
Influence of Maternal Aging on Mitochondrial Heterogeneity, Inheritance, and Function in Oocytes and Preimplantation Embryos.Genes (Basel). 2018 May 21;9(5):265. doi: 10.3390/genes9050265. Genes (Basel). 2018. PMID: 29883421 Free PMC article. Review.
-
Expression of LINE-1 elements is required for preimplantation development and totipotency.Genes Dis. 2025 Feb 7;12(5):101555. doi: 10.1016/j.gendis.2025.101555. eCollection 2025 Sep. Genes Dis. 2025. PMID: 40600062 Free PMC article. Review.
-
Selective Inhibition of mTORC1 Signaling Supports the Development and Maintenance of Pluripotency.Stem Cells. 2024 Jan 13;42(1):13-28. doi: 10.1093/stmcls/sxad079. Stem Cells. 2024. PMID: 37931173 Free PMC article.
-
The role of lipids in genome integrity and pluripotency.Biochem Soc Trans. 2024 Apr 24;52(2):639-650. doi: 10.1042/BST20230479. Biochem Soc Trans. 2024. PMID: 38506536 Free PMC article. Review.
References
-
- Aiken CE, Swoboda PP, Skepper JN, Johnson MH. 2004. The direct measurement of embryogenic volume and nucleo-cytoplasmic ratio during mouse pre-implantation development. Reproduction 128, 527–535. (doi:10.1530/rep.1.00281) - DOI - PubMed
-
- Johnson MH, McConnell J, Van Blerkom J. 1984. Programmed development in the mouse embryo. J. Embryol. Exp. Morphol. 83, 197–231. - PubMed
-
- Morris SA, Guo Y, Zernicka-Goetz M. 2012. Developmental plasticity is bound by pluripotency and the Fgf and Wnt signaling pathways. Cell Rep. 2, 756–765. (doi:10.1016/j.celrep.2012.08.029) - DOI - PMC - PubMed
-
- Artus J, Cohen-Tannoudji M. 2008. Cell cycle regulation during early mouse embryogenesis. Mol. Cell. Endocrinol. 282, 78–86. (doi:10.1016/j.mce.2007.11.008) - DOI - PubMed
-
- Aoki F, Worrad DM, Schultz RM. 1997. Regulation of transcriptional activity during the first and second cell cycles in the preimplantation mouse embryo. Dev. Biol. 181, 296–307. (doi:10.1006/dbio.1996.8466) - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

