Iron Induces Anti-tumor Activity in Tumor-Associated Macrophages
- PMID: 29167669
- PMCID: PMC5682327
- DOI: 10.3389/fimmu.2017.01479
Iron Induces Anti-tumor Activity in Tumor-Associated Macrophages
Abstract
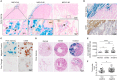
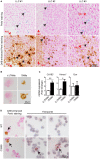
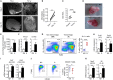
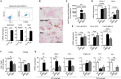
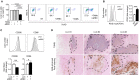
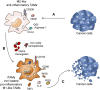
Tumor-associated macrophages (TAMs) frequently help to sustain tumor growth and mediate immune suppression in the tumor microenvironment (TME). Here, we identified a subset of iron-loaded, pro-inflammatory TAMs localized in hemorrhagic areas of the TME. The occurrence of iron-loaded TAMs (iTAMs) correlated with reduced tumor size in patients with non-small cell lung cancer. Ex vivo experiments established that TAMs exposed to hemolytic red blood cells (RBCs) were converted into pro-inflammatory macrophages capable of directly killing tumor cells. This anti-tumor effect could also be elicited via iron oxide nanoparticles. When tested in vivo, tumors injected with such iron oxide nanoparticles led to significantly smaller tumor sizes compared to controls. These results identify hemolytic RBCs and iron as novel players in the TME that repolarize TAMs to exert direct anti-tumor effector function. Thus, the delivery of iron to TAMs emerges as a simple adjuvant therapeutic strategy to promote anti-cancer immune responses.
Keywords: anti-tumor activity; heme; hemolytic red blood cells; iron; iron nanoparticles; macrophage polarization; non-small cell lung cancer; tumor-associated macrophages.
Figures

Similar articles
-
Functional 2D Iron-Based Nanosheets for Synergistic Immunotherapy, Phototherapy, and Chemotherapy of Tumor.Adv Healthc Mater. 2022 Oct;11(19):e2200776. doi: 10.1002/adhm.202200776. Epub 2022 Aug 12. Adv Healthc Mater. 2022. PMID: 35912918
-
Iron accumulation in tumor-associated macrophages marks an improved overall survival in patients with lung adenocarcinoma.Sci Rep. 2019 Aug 5;9(1):11326. doi: 10.1038/s41598-019-47833-x. Sci Rep. 2019. PMID: 31383898 Free PMC article.
-
Tasquinimod triggers an early change in the polarization of tumor associated macrophages in the tumor microenvironment.J Immunother Cancer. 2015 Dec 15;3:53. doi: 10.1186/s40425-015-0098-5. eCollection 2015. J Immunother Cancer. 2015. PMID: 26673090 Free PMC article.
-
Epigenetic control of macrophage polarization: implications for targeting tumor-associated macrophages.Oncotarget. 2018 Feb 21;9(29):20908-20927. doi: 10.18632/oncotarget.24556. eCollection 2018 Apr 17. Oncotarget. 2018. PMID: 29755698 Free PMC article. Review.
-
Tumor-associated macrophages: from basic research to clinical application.J Hematol Oncol. 2017 Feb 28;10(1):58. doi: 10.1186/s13045-017-0430-2. J Hematol Oncol. 2017. PMID: 28241846 Free PMC article. Review.
Cited by
-
Differently Charged Super-Paramagnetic Iron Oxide Nanoparticles Preferentially Induced M1-Like Phenotype of Macrophages.Front Bioeng Biotechnol. 2020 May 29;8:537. doi: 10.3389/fbioe.2020.00537. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 32548111 Free PMC article.
-
The role of iron homeostasis and iron-mediated ROS in cancer.Am J Cancer Res. 2021 May 15;11(5):1895-1912. eCollection 2021. Am J Cancer Res. 2021. PMID: 34094660 Free PMC article. Review.
-
Enhancing cancer immunotherapy: Nanotechnology-mediated immunotherapy overcoming immunosuppression.Acta Pharm Sin B. 2024 Sep;14(9):3834-3854. doi: 10.1016/j.apsb.2024.05.032. Epub 2024 Jun 3. Acta Pharm Sin B. 2024. PMID: 39309502 Free PMC article. Review.
-
ENO1 suppresses cancer cell ferroptosis by degrading the mRNA of iron regulatory protein 1.Nat Cancer. 2022 Jan;3(1):75-89. doi: 10.1038/s43018-021-00299-1. Epub 2021 Dec 9. Nat Cancer. 2022. PMID: 35121990
-
Monitoring innate immune cell dynamics in the glioma microenvironment by magnetic resonance imaging and multiphoton microscopy (MR-MPM).Theranostics. 2020 Jan 1;10(4):1873-1883. doi: 10.7150/thno.38659. eCollection 2020. Theranostics. 2020. PMID: 32042342 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

