TGFβ as a therapeutic target in cystic fibrosis
- PMID: 29168406
- PMCID: PMC6094931
- DOI: 10.1080/14728222.2018.1406922
TGFβ as a therapeutic target in cystic fibrosis
Abstract
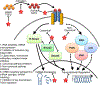
Cystic fibrosis (CF) is a genetic disease characterized by progressive lung disease. Most CF therapies focus on treating secondary pulmonary complications rather than addressing the underlying processes inducing airway remodeling and ineffective response to infection. Transforming growth factor beta (TGFβ) is a cytokine involved in fibrosis, inflammation, and injury response as well as a genetic modifier and biomarker of CF lung disease. Targeting the TGFβ pathway has been pursued in other diseases, but the mechanism of TGFβ effects in CF is less well understood. Areas covered: In this review, we discuss CF lung disease pathogenesis with a focus on potential links to TGFβ. TGFβ signaling in lung health and disease is reviewed. Recent studies investigating TGFβ's impact in CF airway epithelial cells are highlighted. Finally, an overview of potential therapies to target TGFβ signaling relevant to CF are addressed. Expert opinion: The broad impact of TGFβ signaling on numerous cellular processes in homeostasis and disease is both a strength and a challenge to developing TGFβ dependent therapeutics in CF. We discuss the challenges inherent in developing TGFβ-targeted therapy, identifying appropriate patient populations, and questions regarding the timing of treatment. Future directions for research into TGFβ focused therapeutics are discussed.
Keywords: Cystic fibrosis; cystic fibrosis transmembrane conductance regulator; genetic modifier; transforming growth factor beta.
Conflict of interest statement
Declaration of Interest
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Peer reviewers on this manuscript have no relevant financial or other relationships to disclose
Figures
Similar articles
-
Subacute TGFβ Exposure Drives Airway Hyperresponsiveness in Cystic Fibrosis Mice through the PI3K Pathway.Am J Respir Cell Mol Biol. 2020 May;62(5):657-667. doi: 10.1165/rcmb.2019-0158OC. Am J Respir Cell Mol Biol. 2020. PMID: 31922900 Free PMC article.
-
Subacute TGFβ expression drives inflammation, goblet cell hyperplasia, and pulmonary function abnormalities in mice with effects dependent on CFTR function.Am J Physiol Lung Cell Mol Physiol. 2018 Sep 1;315(3):L456-L465. doi: 10.1152/ajplung.00530.2017. Epub 2018 Jun 7. Am J Physiol Lung Cell Mol Physiol. 2018. PMID: 29877096 Free PMC article.
-
CFTR dysfunction in smooth muscle drives TGFβ dependent airway hyperreactivity.Respir Res. 2023 Aug 11;24(1):198. doi: 10.1186/s12931-023-02495-2. Respir Res. 2023. PMID: 37568151 Free PMC article.
-
The epithelial sodium channel (ENaC) as a therapeutic target for cystic fibrosis lung disease.Expert Opin Ther Targets. 2018 Aug;22(8):687-701. doi: 10.1080/14728222.2018.1501361. Epub 2018 Jul 26. Expert Opin Ther Targets. 2018. PMID: 30028216 Review.
-
An update on new and emerging therapies for cystic fibrosis.Expert Opin Emerg Drugs. 2017 Dec;22(4):331-346. doi: 10.1080/14728214.2017.1418324. Epub 2017 Dec 22. Expert Opin Emerg Drugs. 2017. PMID: 29264936 Review.
Cited by
-
Oxy210, a Semi-Synthetic Oxysterol, Inhibits Profibrotic Signaling in Cellular Models of Lung and Kidney Fibrosis.Pharmaceuticals (Basel). 2023 Jan 12;16(1):114. doi: 10.3390/ph16010114. Pharmaceuticals (Basel). 2023. PMID: 36678611 Free PMC article.
-
Antifibrotic mechanism of avitinib in bleomycin-induced pulmonary fibrosis in mice.BMC Pulm Med. 2023 Mar 22;23(1):94. doi: 10.1186/s12890-023-02385-9. BMC Pulm Med. 2023. PMID: 36949426 Free PMC article.
-
Subacute TGFβ Exposure Drives Airway Hyperresponsiveness in Cystic Fibrosis Mice through the PI3K Pathway.Am J Respir Cell Mol Biol. 2020 May;62(5):657-667. doi: 10.1165/rcmb.2019-0158OC. Am J Respir Cell Mol Biol. 2020. PMID: 31922900 Free PMC article.
-
Burkholderia cepacia in cystic fibrosis children and adolescents: overall survival and immune alterations.Front Cell Infect Microbiol. 2024 Jul 1;14:1374318. doi: 10.3389/fcimb.2024.1374318. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39011515 Free PMC article.
-
Human epididymis protein 4 (HE4) protects against cystic pulmonary fibrosis associated-inflammation through inhibition of NF-κB and MAPK singnaling.Genes Genomics. 2019 Sep;41(9):1045-1053. doi: 10.1007/s13258-019-00836-4. Epub 2019 Jun 4. Genes Genomics. 2019. PMID: 31165362
References
-
- North American Cystic Fibrosis Foundation. Patient Registry: Annual Data Report. 2015. Available from: https://www.cff.org/Our-Research/CF-Patient-Registry/2015-Patient-Regist...
-
- Rowe SM, Miller S, Sorscher EJ. Cystic fibrosis. N Engl J Med. 2005;352:1992–2001 - PubMed
-
-
Sly PD, Gangell CL, Chen L, et al. Risk factors for bronchiectasis in children with cystic fibrosis. N Engl J Med. 2013;368:1963–1970.
Description of early lung disease in children with CF under age 3 years.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical