Study design considerations for the Standardized Treatment of Pulmonary Exacerbations 2 (STOP2): A trial to compare intravenous antibiotic treatment durations in CF
- PMID: 29170074
- PMCID: PMC5742044
- DOI: 10.1016/j.cct.2017.11.012
Study design considerations for the Standardized Treatment of Pulmonary Exacerbations 2 (STOP2): A trial to compare intravenous antibiotic treatment durations in CF
Abstract
Background: Pulmonary exacerbations (PEx) in cystic fibrosis (CF) are common and contribute to morbidity and mortality. Duration of IV antibiotic therapy to treat PEx varies widely in the US, and there are few data to guide treatment decisions.
Methods: We combined a survey of CF stakeholders with retrospective analyses of a recent observational study of CF PEx to design a multicenter, randomized, prospective study comparing the efficacy and safety of different durations of IV antibiotics for PEx to meet the needs of people with CF and their caregivers.
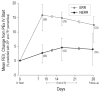
Results: IV antibiotic duration was cited as the most important PEx research question by responding CF physicians and top concern among surveyed CF patients/caregivers. During PEx, forced expiratory volume in 1s (FEV1% predicted) and symptom responses at 7-10days of IV antibiotics identified two distinct groups: early robust responders (ERR) who subsequently experienced greater FEV1 improvements compared to non-ERR (NERR). In addition to greater FEV1 and symptom responses, only 14% of ERR patients were treated with IV antibiotics for >15days, compared with 45% of NERR patients.
Conclusions: A divergent trial design that evaluates subjects' interim improvement in FEV1 and symptoms to tailor randomization to IV treatment duration (10 vs. 14days for ERR, 14 vs. 21days for NERR) may alleviate physician and patient concerns about excess or inadequate treatment. Such a study has the potential to provide evidence necessary to standardize IV antibiotic duration in CF PEx care -a first step to conducting PEx research of other treatment features.
Keywords: Cystic fibrosis; FEV1; Pulmonary exacerbation; Symptoms.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures



References
-
- Konstan MW, Morgan WJ, Butler SM, Pasta DJ, Craib ML, Silva SJ, Stokes DC, Wohl ME, Wagener JS, Regelmann WE, Johnson CA Scientific Advisory Group and the Investigators and Coordinators of the Epidemiologic Study of Cystic Fibrosis. Risk factors for rate of decline in forced expiratory volume in one second in children and adolescents with cystic fibrosis. J Pediatr. 2007;151:134–139. - PubMed
-
- Waters V, Stanojevic S, Atenafu EG, Lu A, Yau Y, Tullis E, Ratjen F. Effect of pulmonary exacerbations on long-term lung function decline in cystic fibrosis. Eur Respir J. 2012;40:61–6. - PubMed
-
- de Boer K, Vandemheen KL, Tullis E, Doucette S, Fergusson D, Freitag A, Paterson N, Jackson M, Lougheed MD, Kumar V, Aaron SD. Exacerbation frequency and clinical outcomes in adult patients with cystic fibrosis. Thorax. 2011;66:680–5. - PubMed
-
- Cystic Fibrosis Foundation Patient Registry: Annual Data Report 2015. Bethesda, MD: Cystic Fibrosis Foundation; 2015.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

