Stress-responsive FKBP51 regulates AKT2-AS160 signaling and metabolic function
- PMID: 29170369
- PMCID: PMC5700978
- DOI: 10.1038/s41467-017-01783-y
Stress-responsive FKBP51 regulates AKT2-AS160 signaling and metabolic function
Abstract
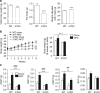
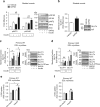
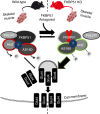
The co-chaperone FKBP5 is a stress-responsive protein-regulating stress reactivity, and its genetic variants are associated with T2D related traits and other stress-related disorders. Here we show that FKBP51 plays a role in energy and glucose homeostasis. Fkbp5 knockout (51KO) mice are protected from high-fat diet-induced weight gain, show improved glucose tolerance and increased insulin signaling in skeletal muscle. Chronic treatment with a novel FKBP51 antagonist, SAFit2, recapitulates the effects of FKBP51 deletion on both body weight regulation and glucose tolerance. Using shorter SAFit2 treatment, we show that glucose tolerance improvement precedes the reduction in body weight. Mechanistically, we identify a novel association between FKBP51 and AS160, a substrate of AKT2 that is involved in glucose uptake. FKBP51 antagonism increases the phosphorylation of AS160, increases glucose transporter 4 expression at the plasma membrane, and ultimately enhances glucose uptake in skeletal myotubes. We propose FKBP51 as a mediator between stress and T2D development, and potential target for therapeutic approaches.
Conflict of interest statement
The authors declare no competing financial interests.
Figures




Similar articles
-
Rab GAPs AS160 and Tbc1d1 play nonredundant roles in the regulation of glucose and energy homeostasis in mice.Am J Physiol Endocrinol Metab. 2016 Feb 15;310(4):E276-88. doi: 10.1152/ajpendo.00342.2015. Epub 2015 Dec 1. Am J Physiol Endocrinol Metab. 2016. PMID: 26625902 Free PMC article.
-
Tilorone increases glucose uptake in vivo and in skeletal muscle cells by enhancing Akt2/AS160 signaling and glucose transporter levels.J Cell Physiol. 2023 May;238(5):1080-1094. doi: 10.1002/jcp.30998. Epub 2023 Apr 3. J Cell Physiol. 2023. PMID: 37012691
-
Whole body glucoregulation and tissue-specific glucose uptake in a novel Akt substrate of 160 kDa knockout rat model.PLoS One. 2019 Apr 29;14(4):e0216236. doi: 10.1371/journal.pone.0216236. eCollection 2019. PLoS One. 2019. PMID: 31034517 Free PMC article.
-
Role of Akt substrate of 160 kDa in insulin-stimulated and contraction-stimulated glucose transport.Appl Physiol Nutr Metab. 2007 Jun;32(3):557-66. doi: 10.1139/H07-026. Appl Physiol Nutr Metab. 2007. PMID: 17510697 Review.
-
Focus on FKBP51: A molecular link between stress and metabolic disorders.Mol Metab. 2019 Nov;29:170-181. doi: 10.1016/j.molmet.2019.09.003. Epub 2019 Sep 11. Mol Metab. 2019. PMID: 31668388 Free PMC article. Review.
Cited by
-
Proof-of-concept for CRISPR/Cas9 gene editing in human preadipocytes: Deletion of FKBP5 and PPARG and effects on adipocyte differentiation and metabolism.Sci Rep. 2020 Jun 29;10(1):10565. doi: 10.1038/s41598-020-67293-y. Sci Rep. 2020. PMID: 32601291 Free PMC article.
-
Downregulation of FKBP5 Promotes Atrial Arrhythmogenesis.Circ Res. 2023 Jun 23;133(1):e1-e16. doi: 10.1161/CIRCRESAHA.122.322213. Epub 2023 May 8. Circ Res. 2023. PMID: 37154033 Free PMC article.
-
Stress and glucocorticoid modulation of feeding and metabolism.Neurobiol Stress. 2019 May 15;11:100171. doi: 10.1016/j.ynstr.2019.100171. eCollection 2019 Nov. Neurobiol Stress. 2019. PMID: 31193462 Free PMC article. Review.
-
Deep phenotyping reveals CRH and FKBP51-dependent behavioral profiles following chronic social stress exposure in male mice.Neuropsychopharmacology. 2025 Feb;50(3):556-567. doi: 10.1038/s41386-024-02008-9. Epub 2024 Oct 22. Neuropsychopharmacology. 2025. PMID: 39438757 Free PMC article.
-
Large-scale, in-cell photocrosslinking at single-residue resolution reveals the molecular basis for glucocorticoid receptor regulation by immunophilins.Nat Struct Mol Biol. 2023 Dec;30(12):1857-1866. doi: 10.1038/s41594-023-01098-1. Epub 2023 Nov 9. Nat Struct Mol Biol. 2023. PMID: 37945739
References
-
- Vermeer H, Hendriks-Stegeman BI, van der Burg B, van Buul-Offers SC, Jansen M. Glucocorticoid-induced increase in lymphocytic {FKBP}51 messenger ribonucleic acid expression: a potential marker for glucocorticoid sensitivity, potency, and bioavailability. J. Clin. Endocrinol. Metab. 2003;88:277–284. doi: 10.1210/jc.2002-020354. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

