Anthocyanins from the Fruit of Vitis Coignetiae Pulliat Inhibit TNF-Augmented Cancer Proliferation, Migration, and Invasion in A549 Cells
- PMID: 29172259
- PMCID: PMC5773771
- DOI: 10.22034/APJCP.2017.18.11.2919
Anthocyanins from the Fruit of Vitis Coignetiae Pulliat Inhibit TNF-Augmented Cancer Proliferation, Migration, and Invasion in A549 Cells
Abstract
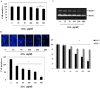
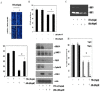
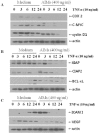
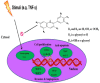
Objective: Anthocyanins belong to a class of flavonoids, exhibiting antioxidant and anti-inflammatory actions have been reported to have anti-cancer effects. Here, we investigated whether anthocyanins can inhibit cancer cell proliferation, invasion, and angiogenesis in human lung cancer A549 cells, which are critically involved in cancer metastasis. Methods: We used anthocyanins from fruits of Vitis coignetiae Pulliat (AIMs) which has been used in Korean folk medicine for the treatment of inflammatory diseases and cancers. We have performed cell proliferation assays, cell invasion assay, gelatin zymography, wound healing assay and western blotting to examine whether anthocyanins can inhibit cancer cell proliferation, invasion, and angiogenesis in A549 cells. Result: AIMs did not inhibit cancer cell proliferation on A549 cells. Also, AIMs suppressed cancer migration, and invasion by supressing MMP-2 and MMP-9 expression. The Immuno-blotting results also revealed that AIMs suppressed the proteins involved in cancer proliferation (COX- 2, C-myc, cyclin D1), migration and invasion (MMP-2, MMP-9), anti-apoptosis (XIAP, and c-IAP2), adhesion and angiogenesis (ICAM-1, VEGF). Conclusion: This study demonstrates that the anthocyanins isolated from fruits of Vitis coignetiae Pulliat inhibit cancer proliferation, cancer migration, and invasion that is involve in cancer-metastasis. This study provides evidence that AIMs might have anti-cancer effects on human lung cancer.
Keywords: Anthocyanins; Vitis coignetiae Pulliat; invasion; anti-cancer effects; lung cancer.
Creative Commons Attribution License
Figures

Similar articles
-
Insights into the Mechanisms of Action of Proanthocyanidins and Anthocyanins in the Treatment of Nicotine-Induced Non-Small Cell Lung Cancer.Int J Mol Sci. 2022 Jul 18;23(14):7905. doi: 10.3390/ijms23147905. Int J Mol Sci. 2022. PMID: 35887251 Free PMC article. Review.
-
Pretreatment of Anthocyanin from the Fruit of Vitis coignetiae Pulliat Acts as a Potent Inhibitor of TNF-α Effect by Inhibiting NF-κB-Regulated Genes in Human Breast Cancer Cells.Molecules. 2020 May 21;25(10):2396. doi: 10.3390/molecules25102396. Molecules. 2020. PMID: 32455624 Free PMC article.
-
Characterization of a profile of the anthocyanins isolated from Vitis coignetiae Pulliat and their anti-invasive activity on HT-29 human colon cancer cells.Food Chem Toxicol. 2010 Mar;48(3):903-9. doi: 10.1016/j.fct.2009.12.031. Epub 2010 Jan 6. Food Chem Toxicol. 2010. PMID: 20060025
-
Anthocyanins From the Fruit of Vitis coignetiae Pulliat Potentiate the Cisplatin Activity by Inhibiting PI3K/Akt Signaling Pathways in Human Gastric Cancer Cells.J Cancer Prev. 2015 Mar;20(1):50-6. doi: 10.15430/JCP.2015.20.1.50. J Cancer Prev. 2015. PMID: 25853103 Free PMC article.
-
Anthocyanins Derived from Vitis coignetiae Pulliat Contributes Anti-Cancer Effects by Suppressing NF-κB Pathways in Hep3B Human Hepatocellular Carcinoma Cells and In Vivo.Molecules. 2020 Nov 20;25(22):5445. doi: 10.3390/molecules25225445. Molecules. 2020. PMID: 33233701 Free PMC article.
Cited by
-
Bioactive Compounds: Natural Defense Against Cancer?Biomolecules. 2019 Nov 21;9(12):758. doi: 10.3390/biom9120758. Biomolecules. 2019. PMID: 31766399 Free PMC article. Review.
-
Insights into the Mechanisms of Action of Proanthocyanidins and Anthocyanins in the Treatment of Nicotine-Induced Non-Small Cell Lung Cancer.Int J Mol Sci. 2022 Jul 18;23(14):7905. doi: 10.3390/ijms23147905. Int J Mol Sci. 2022. PMID: 35887251 Free PMC article. Review.
-
Behind the Scenes of Anthocyanins-From the Health Benefits to Potential Applications in Food, Pharmaceutical and Cosmetic Fields.Nutrients. 2022 Dec 2;14(23):5133. doi: 10.3390/nu14235133. Nutrients. 2022. PMID: 36501163 Free PMC article. Review.
-
Anthocyanins, Vibrant Color Pigments, and Their Role in Skin Cancer Prevention.Biomedicines. 2020 Sep 9;8(9):336. doi: 10.3390/biomedicines8090336. Biomedicines. 2020. PMID: 32916849 Free PMC article. Review.
-
Anthocyanins: From Natural Colorants to Potent Anticancer Agents.Food Sci Nutr. 2025 May 2;13(5):e70232. doi: 10.1002/fsn3.70232. eCollection 2025 May. Food Sci Nutr. 2025. PMID: 40321606 Free PMC article. Review.
References
-
- Aggarwal BB. Nuclear factor-kappaB: the enemy within. Cancer Cell. 2004;6:203–8. - PubMed
-
- Aggarwal S, Ichikawa H, Takada Y, et al. Curcumin (diferuloylmethane) down-regulates expression of cell proliferation and antiapoptotic and metastatic gene products through suppression of IkappaBalpha kinase and Akt activation. Mol Pharmacol. 2006;69:195–206. - PubMed
-
- Balkwill F. TNF-alpha in promotion and progression of cancer. Cancer Metastasis Rev. 2006;25:409–16. - PubMed
-
- Chun KH, Kosmeder JW, Sun S, et al. Effects of deguelin on the phosphatidylinositol 3-kinase/Akt pathway and apoptosis in premalignant human bronchial epithelial cells. J Natl Cancer Inst. 2003;95:291–302. - PubMed
-
- Favot L, Martin S, Keravis T, et al. Involvement of cyclin-dependent pathway in the inhibitory effect of delphinidin on angiogenesis. Cardiovasc Res. 2003;59:479–87. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

