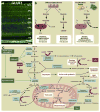
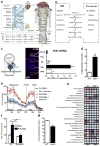
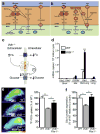
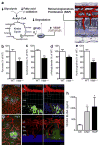
Retinal energy demands control vascular supply of the retina in development and disease: The role of neuronal lipid and glucose metabolism
- PMID: 29175509
- PMCID: PMC5963988
- DOI: 10.1016/j.preteyeres.2017.11.002
Retinal energy demands control vascular supply of the retina in development and disease: The role of neuronal lipid and glucose metabolism
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

