Oral versus intravenous methylprednisolone for the treatment of multiple sclerosis relapses: A meta-analysis of randomized controlled trials
- PMID: 29176905
- PMCID: PMC5703548
- DOI: 10.1371/journal.pone.0188644
Oral versus intravenous methylprednisolone for the treatment of multiple sclerosis relapses: A meta-analysis of randomized controlled trials
Abstract
Background: Intravenous glucocorticoids are recommended for multiple sclerosis (MS). However, they can be inconvenient and expensive. Due to their convenience and low cost, oral glucocorticoids may be an alternative treatment. Recently, several studies have shown that there is no difference in efficacy and safety between oral methylprednisolone (oMP) and intravenous methylprednisolone (ivMP).
Objectives: We sought to assess the clinical efficacy, safety and tolerability of oral methylprednisolone versus intravenous methylprednisolone for MS relapses in this meta-analysis.
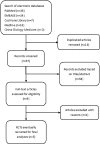
Methods: Randomized controlled trials (RCTs) evaluating the clinical efficacy, safety and tolerability of oral methylprednisolone versus intravenous methylprednisolone for MS relapses were searched in PubMed, Cochrane Library, Medline, EMBASE and China Biology Medicine until October 25, 2016, without language restrictions. The proportion of patients who had improved by day 28 was chosen as the efficacy outcome. We chose the risk ratio (RR) to analyze each trial with the 95% confidence interval (95% CI). We also used the fixed-effects model (Mantel-Haenszel approach) to calculate the pooled relative effect estimates.
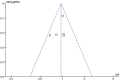
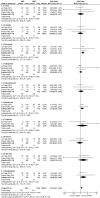
Results: A total of 5 trials were identified, which included 369 patients. The results of our meta-analysis revealed that no significant difference existed in relapse improvement at day 28 between oMP and ivMP (RR 0.96, 95% CI 0.84 to 1.10). No evidence of heterogeneity existed among the trials (P = 0.45, I2 = 0%). Both treatments were equally safe and well tolerated except that insomnia was more likely to occur in the oMP group compared to the ivMP group.
Conclusion: Our meta-analysis reveals strong evidence that oMP is not inferior to ivMP in increasing the proportion of patients experiencing clinical improvement at day 28. In addition, both routes of administration are equally well tolerated and safe. These findings suggest that we may be able to replace ivMP with oMP to treat MS relapses.
Conflict of interest statement
Figures
Similar articles
-
A randomized clinical trial of oral versus intravenous methylprednisolone for relapse of MS.Mult Scler. 2014 May;20(6):717-25. doi: 10.1177/1352458513508835. Epub 2013 Oct 21. Mult Scler. 2014. PMID: 24144876 Clinical Trial.
-
Similar biological effect of high-dose oral versus intravenous methylprednisolone in multiple sclerosis relapses.Mult Scler. 2015 Apr;21(5):646-50. doi: 10.1177/1352458514546786. Epub 2014 Aug 21. Mult Scler. 2015. PMID: 25145693 Clinical Trial.
-
A short-term randomized MRI study of high-dose oral vs intravenous methylprednisolone in MS.Neurology. 2009 Dec 1;73(22):1842-8. doi: 10.1212/WNL.0b013e3181c3fd5b. Neurology. 2009. PMID: 19949030 Clinical Trial.
-
Re-examining the effects of high-dose intravenous methylprednisolone for secondary progressive multiple sclerosis.Neurodegener Dis Manag. 2021 Apr;11(2):177-185. doi: 10.2217/nmt-2020-0051. Epub 2021 Mar 11. Neurodegener Dis Manag. 2021. PMID: 33703936
-
Would it be recommended treating multiple sclerosis relapses with high dose oral instead intravenous steroids during the COVID-19 pandemic? Yes.Mult Scler Relat Disord. 2020 Nov;46:102449. doi: 10.1016/j.msard.2020.102449. Epub 2020 Aug 20. Mult Scler Relat Disord. 2020. PMID: 32853893 Free PMC article. Review.
Cited by
-
Treatment Effectiveness for Resolution of Multiple Sclerosis Relapse in a US Health Plan Population.Neurol Ther. 2019 Dec;8(2):383-395. doi: 10.1007/s40120-019-00156-5. Epub 2019 Sep 28. Neurol Ther. 2019. PMID: 31564036 Free PMC article.
-
Horizontal Gaze Palsy and Ipsilateral Facial Nerve Palsy in Older Patient as Initial Manifestation of Very Late-Onset Multiple Sclerosis Successfully Treated with Oral Corticosteroids: A Case Report.Case Rep Ophthalmol. 2024 Mar 5;15(1):176-183. doi: 10.1159/000536639. eCollection 2024 Jan-Dec. Case Rep Ophthalmol. 2024. PMID: 38444523 Free PMC article.
-
Intranasal Methylprednisolone Ameliorates Neuroinflammation Induced by Chronic Toluene Exposure.Pharmaceutics. 2022 Jun 2;14(6):1195. doi: 10.3390/pharmaceutics14061195. Pharmaceutics. 2022. PMID: 35745768 Free PMC article.
-
Approach to Managing the Initial Presentation of Multiple Sclerosis: A Worldwide Practice Survey.Neurol Clin Pract. 2025 Feb;15(1):e200376. doi: 10.1212/CPJ.0000000000200376. Epub 2024 Oct 9. Neurol Clin Pract. 2025. PMID: 39399557
-
Update on the management of multiple sclerosis during the COVID-19 pandemic and post pandemic: An international consensus statement.J Neuroimmunol. 2021 Aug 15;357:577627. doi: 10.1016/j.jneuroim.2021.577627. Epub 2021 Jun 7. J Neuroimmunol. 2021. PMID: 34139567 Free PMC article. Review.
References
-
- Noseworthy JH, Lucchinetti C, Rodriguez M, Weinshenker BG (2000) Multiple sclerosis. N Engl J Med 343: 938–952. doi: 10.1056/NEJM200009283431307 - DOI - PubMed
-
- Ramagopalan SV, Sadovnick AD (2011) Epidemiology of multiple sclerosis. Neurol Clin 29: 207–217. doi: 10.1016/j.ncl.2010.12.010 - DOI - PubMed
-
- Lublin FD, Reingold SC (1996) Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology 46: 907–911. - PubMed
-
- Lublin FD, Reingold SC, Cohen JA, Cutter GR, Sorensen PS, Thompson AJ, et al. (2014) Defining the clinical course of multiple sclerosis: the 2013 revisions. Neurology 83: 278–286. doi: 10.1212/WNL.0000000000000560 - DOI - PMC - PubMed
-
- Weinshenker BG (1994) Natural history of multiple sclerosis. Ann Neurol 36 Suppl: S6–11. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical