Spinal Cord Injury Disrupts Resting-State Networks in the Human Brain
- PMID: 29179629
- PMCID: PMC5863102
- DOI: 10.1089/neu.2017.5212
Spinal Cord Injury Disrupts Resting-State Networks in the Human Brain
Abstract
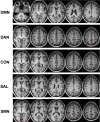
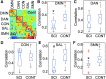
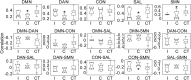
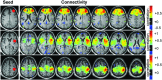
Despite 253,000 spinal cord injury (SCI) patients in the United States, little is known about how SCI affects brain networks. Spinal MRI provides only structural information with no insight into functional connectivity. Resting-state functional MRI (RS-fMRI) quantifies network connectivity through the identification of resting-state networks (RSNs) and allows detection of functionally relevant changes during disease. Given the robust network of spinal cord afferents to the brain, we hypothesized that SCI produces meaningful changes in brain RSNs. RS-fMRIs and functional assessments were performed on 10 SCI subjects. Blood oxygen-dependent RS-fMRI sequences were acquired. Seed-based correlation mapping was performed using five RSNs: default-mode (DMN), dorsal-attention (DAN), salience (SAL), control (CON), and somatomotor (SMN). RSNs were compared with normal control subjects using false-discovery rate-corrected two way t tests. SCI reduced brain network connectivity within the SAL, SMN, and DMN and disrupted anti-correlated connectivity between CON and SMN. When divided into separate cohorts, complete but not incomplete SCI disrupted connectivity within SAL, DAN, SMN and DMN and between CON and SMN. Finally, connectivity changed over time after SCI: the primary motor cortex decreased connectivity with the primary somatosensory cortex, the visual cortex decreased connectivity with the primary motor cortex, and the visual cortex decreased connectivity with the sensory parietal cortex. These unique findings demonstrate the functional network plasticity that occurs in the brain as a result of injury to the spinal cord. Connectivity changes after SCI may serve as biomarkers to predict functional recovery following an SCI and guide future therapy.
Keywords: MRI; SCI; biomarkers; neuroplasticity.
Conflict of interest statement
No competing financial interests exist.
Figures

Similar articles
-
Alteration of Resting-State Brain Sensorimotor Connectivity following Spinal Cord Injury: A Resting-State Functional Magnetic Resonance Imaging Study.J Neurotrauma. 2015 Sep 15;32(18):1422-7. doi: 10.1089/neu.2014.3661. Epub 2015 Jun 25. J Neurotrauma. 2015. PMID: 25945389
-
Cortical plasticity after brachial plexus injury and repair: a resting-state functional MRI study.Neurosurg Focus. 2017 Mar;42(3):E14. doi: 10.3171/2016.12.FOCUS16430. Neurosurg Focus. 2017. PMID: 28245732
-
Alterations of resting-state regional and network-level neural function after acute spinal cord injury.Neuroscience. 2014 Sep 26;277:446-54. doi: 10.1016/j.neuroscience.2014.07.045. Epub 2014 Jul 30. Neuroscience. 2014. PMID: 25086312
-
Resting-state network alterations in depression: a comprehensive meta-analysis of functional connectivity.Psychol Med. 2025 Feb 26;55:e63. doi: 10.1017/S0033291725000303. Psychol Med. 2025. PMID: 40008424 Free PMC article.
-
Opposite effects of dopamine and serotonin on resting-state networks: review and implications for psychiatric disorders.Mol Psychiatry. 2020 Jan;25(1):82-93. doi: 10.1038/s41380-019-0406-4. Epub 2019 Apr 5. Mol Psychiatry. 2020. PMID: 30953003 Review.
Cited by
-
A Distance-Based Neurorehabilitation Evaluation Method Using Linear SVM and Resting-State fMRI.Front Neurol. 2019 Nov 1;10:1105. doi: 10.3389/fneur.2019.01105. eCollection 2019. Front Neurol. 2019. PMID: 31736850 Free PMC article.
-
Disentangling the Effects of Spinal Cord Injury and Related Neuropathic Pain on Supraspinal Neuroplasticity: A Systematic Review on Neuroimaging.Front Neurol. 2020 Feb 5;10:1413. doi: 10.3389/fneur.2019.01413. eCollection 2019. Front Neurol. 2020. PMID: 32116986 Free PMC article.
-
Different macaque brain network remodeling after spinal cord injury and NT3 treatment.iScience. 2023 Apr 29;26(6):106784. doi: 10.1016/j.isci.2023.106784. eCollection 2023 Jun 16. iScience. 2023. PMID: 37378337 Free PMC article.
-
Supraspinal nociceptive networks in neuropathic pain after spinal cord injury.Hum Brain Mapp. 2021 Aug 15;42(12):3733-3749. doi: 10.1002/hbm.25401. Epub 2021 Jun 16. Hum Brain Mapp. 2021. PMID: 34132441 Free PMC article.
-
Electroencephalography as a Biomarker for Functional Recovery in Spinal Cord Injury Patients.Front Hum Neurosci. 2021 Apr 9;15:548558. doi: 10.3389/fnhum.2021.548558. eCollection 2021. Front Hum Neurosci. 2021. PMID: 33897390 Free PMC article.
References
-
- Kadanka Z., Bednarik J., Vohanka S., Vlach O., Stejskal L., Chaloupka R., Filipovicova D., Surelova D., Adamova B., Novotny O., Nemec M., Smrcka V., and Urbanek I. (2000). Conservative treatment versus surgery in spondylotic cervical myelopathy: a prospective randomised study. Eur. Spine J. 9, 538–544 - PMC - PubMed
-
- Nagata K., Yoshimura N., Muraki S., Hashizume H., Ishimoto Y., Yamada H., Takiguchi N., Nakagawa Y., Oka H., Kawaguchi H., Nakamura K., Akune T., and Yoshida M. (2012). Prevalence of cervical cord compression and its association with physical performance in a population-based cohort in Japan: The Wakayama Spine Study. Spine 37, 1892–1898 - PubMed
-
- Boogaarts H., and Bartels R.M.A. (2015). Prevalence of cervical spondylotic myelopathy. Eur. Spine J., 24, Suppl. 2, 139–141 - PubMed
-
- Kirshblum S.C., Burns S.P., Biering-Sorensen F., Donovan W., Graves D.E., Jha A., Johansen M., Jones L., Krassioukov A., Mulcahey M.J., Schmidt-Read M., and Waring W. (2011). International standards for neurological classification of spinal cord injury (revised 2011). J. Spinal Cord Med. 34, 535–546 - PMC - PubMed
-
- Biswal B., Yetkin F.Z., Haughton V.M., and Hyde J.S. (1995). Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn. Reson. Med. 34, 537–541 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

