Phase II Study of BGJ398 in Patients With FGFR-Altered Advanced Cholangiocarcinoma
- PMID: 29182496
- PMCID: PMC6075847
- DOI: 10.1200/JCO.2017.75.5009
Phase II Study of BGJ398 in Patients With FGFR-Altered Advanced Cholangiocarcinoma
Abstract
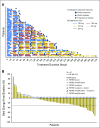
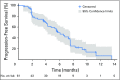
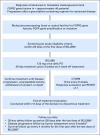
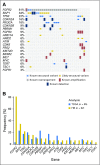
Purpose No standard treatment exists for patients with cholangiocarcinoma for whom first-line gemcitabine-based therapy fails. Fibroblast growth factor receptor 2 ( FGFR2) fusions/translocations are present in 13% to 17% of intrahepatic cholangiocarcinomas. BGJ398, an orally bioavailable, selective pan-FGFR kinase inhibitor, has shown preliminary clinical activity against tumors with FGFR alterations. Methods A multicenter, open-label, phase II study ( ClinicalTrials.gov identifier: NCT02150967) evaluated BGJ398 antitumor activity in patients age ≥ 18 years with advanced or metastatic cholangiocarcinoma containing FGFR2 fusions or other FGFR alterations whose disease had progressed while receiving prior therapy. Patients received BGJ398 125 mg once daily for 21 days, then 7 days off (28-day cycles). The primary end point was investigator-assessed overall response rate. Results Sixty-one patients (35 women; median age, 57 years) with FGFR2 fusion (n = 48), mutation (n = 8), or amplification (n = 3) participated. At the prespecified data cutoff (June 30, 2016), 50 patients had discontinued treatment. All responsive tumors contained FGFR2 fusions. The overall response rate was 14.8% (18.8% FGFR2 fusions only), disease control rate was 75.4% (83.3% FGFR2 fusions only), and estimated median progression-free survival was 5.8 months (95% CI, 4.3 to 7.6 months). Adverse events included hyperphosphatemia (72.1% all grade), fatigue (36.1%), stomatitis (29.5%), and alopecia (26.2%). Grade 3 or 4 treatment-related adverse events occurred in 25 patients (41%) and included hyperphosphatemia (16.4%), stomatitis (6.6%), and palmar-plantar erythrodysesthesia (4.9%). Conclusion BGJ398 is a first-in-class FGFR kinase inhibitor with manageable toxicities that shows meaningful clinical activity against chemotherapy-refractory cholangiocarcinoma containing FGFR2 fusions. This promising antitumor activity supports continued development of BGJ398 in this highly selected patient population.
Figures
Comment in
-
Fibroblast Growth Factor Receptor Inhibition for Cholangiocarcinoma: Looking Through a Door Half-Opened.Hepatology. 2018 Dec;68(6):2428-2430. doi: 10.1002/hep.30069. Epub 2018 Nov 7. Hepatology. 2018. PMID: 29704257 Free PMC article. No abstract available.
References
-
- Alpini G, Prall R, LaRusso NFThe pathobiology of biliary epithelia, in Arias IM, Boyer JL, Chisari FV, et al.: The Liver: Biology and Pathobiology (ed 4). Philadelphia, PA, Lippincott Williams & Wilkins, 2001, pp 421-435
-
- American Cancer Society : Bile duct cancer (cholangiocarcinoma). https://www.cancer.org/cancer/bile-duct-cancer.html
-
- Banales JM, Cardinale V, Carpino G, et al. : Expert consensus document: Cholangiocarcinoma: current knowledge and future perspectives consensus statement from the European Network for the Study of Cholangiocarcinoma (ENS-CCA). Nat Rev Gastroenterol Hepatol 13:261-280, 2016 - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

