The effect of an intensive smoking cessation intervention on disease activity in patients with rheumatoid arthritis: study protocol for a randomised controlled trial
- PMID: 29183347
- PMCID: PMC5706378
- DOI: 10.1186/s13063-017-2309-5
The effect of an intensive smoking cessation intervention on disease activity in patients with rheumatoid arthritis: study protocol for a randomised controlled trial
Abstract
Background: Rheumatoid arthritis (RA) is a chronic, inflammatory rheumatic disease with the potential to induce significant disability. Patients with RA are at increased risk of cardiovascular diseases (CVD). Smokers with RA tend to experience more pain and fatigue, higher disease activity, more erosive joint destruction and a lower health-related quality of life (HR-QoL) than non-smokers. It remains to be determined whether these effects can be reduced by smoking cessation. This randomised controlled trial (RCT) in patients with RA aims to examine the effect of intensive smoking cessation intervention (motivational counselling combined with tailored nicotine replacement therapy) versus standard care on smoking cessation, and consequently on disease activity. Secondary objectives are to explore the effect on flare, risk factors for CVD, lung function, physical function, HR-QoL, pain and fatigue in patients with RA.
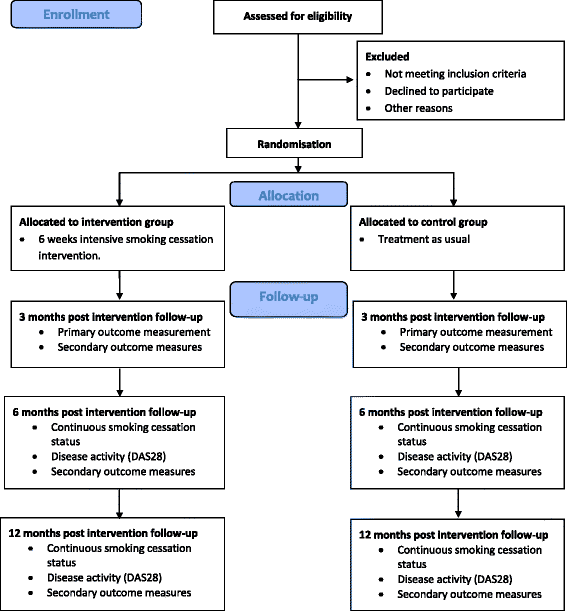
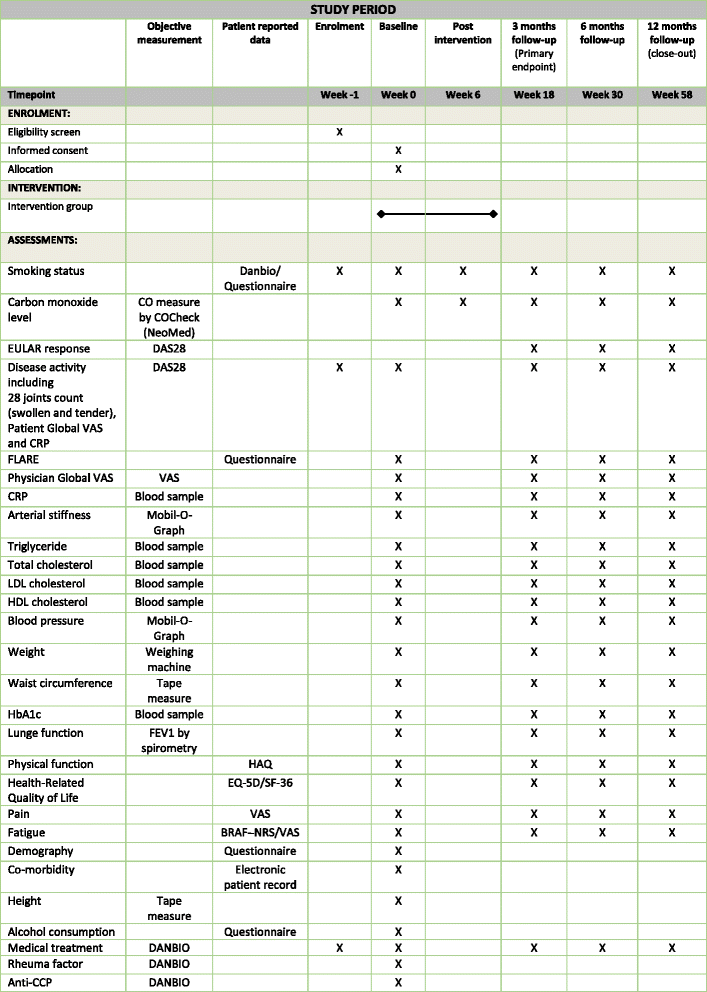
Methods: This will be a multicentre, open label, two arm, parallel group, RCT, including 150 daily smokers with RA, being in remission or having low-moderate disease activity (DAS28 ≤ 5.1). The intervention group (n = 75) will receive five counselling sessions with a trained smoking cessation counsellor based on the principles of motivational counselling. Furthermore, intervention patients will be offered nicotine replacement therapy tailored to individual needs. Participants randomised to the control group will receive standard care. The co-primary outcome is a hierarchical endpoint, which will be evaluated at 3 months follow-up and will include (1) self-reported smoking cessation biochemically validated by exhaled carbon monoxide and (2) achievement of EULAR clinical response (an improvement in DAS28 of > 0.6). Follow-up visits will be performed at 3, 6 and 12 months post-intervention.
Discussion: This trial will reveal whether intensive smoking cessation counselling helps smokers with RA to achieve continuous smoking cessation and whether, as a concomitant benefit, it will reduce their RA disease activity. The trial aims to generate high quality evidence for the feasibility of a health promotion intervention for smokers with RA.
Trial registration: ClinicalTrials.gov, identifier: NCT02901886 . Registered on 10 September 2016. Recruitment status updated on 10th October 2016.
Keywords: Behavioural change; Cardiovascular biomarkers; Cardiovascular disease; Clinical health promotion; Flare; Health-related quality of life; Motivational counselling; Nicotine replacement therapy; Smoking cessation intervention; Tobacco smoking.
Conflict of interest statement
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- Hewlett S, Sanderson T, May J, Alten R, Bingham CO, Cross M, et al. ‘I’m hurting, I want to kill myself’: rheumatoid arthritis flare is more than a high joint count--an international patient perspective on flare where medical help is sought. Rheumatology (Oxford) 2012;51(1):69–76. doi: 10.1093/rheumatology/keq455. - DOI - PubMed
-
- Carr A, Hewlett S, Hughes R, Mitchell H, Ryan S, Carr M, et al. Rheumatology outcomes: the patient’s perspective. J Rheumatol. 2003;30(4):880–3. - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical