Hoxa2 Inhibits Bone Morphogenetic Protein Signaling during Osteogenic Differentiation of the Palatal Mesenchyme
- PMID: 29184513
- PMCID: PMC5694536
- DOI: 10.3389/fphys.2017.00929
Hoxa2 Inhibits Bone Morphogenetic Protein Signaling during Osteogenic Differentiation of the Palatal Mesenchyme
Abstract
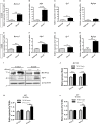
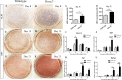
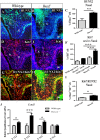
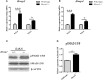
Cleft palate is one of the most common congenital birth defects worldwide. The homeobox (Hox) family of genes are key regulators of embryogenesis, with Hoxa2 having a direct role in secondary palate development. Hoxa2-/- mice exhibit cleft palate; however, the cellular and molecular mechanisms leading to cleft palate in Hoxa2-/- mice is largely unknown. Addressing this issue, we found that Hoxa2 regulates spatial and temporal programs of osteogenic differentiation in the developing palate by inhibiting bone morphogenetic protein (BMP) signaling dependent osteoblast markers. Expression of osteoblast markers, including Runx2, Sp7, and AlpI were increased in Hoxa2-/- palatal shelves at embryonic day (E) 13.5 and E15.5. Hoxa2-/- mouse embryonic palatal mesenchyme (MEPM) cells exhibited increased bone matrix deposition and mineralization in vitro. Moreover, loss of Hoxa2 resulted in increased osteoprogenitor cell proliferation and osteogenic commitment during early stages of palate development at E13.5. Consistent with upregulation of osteoblast markers, Hoxa2-/- palatal shelves displayed higher expression of canonical BMP signaling in vivo. Blocking BMP signaling in Hoxa2-/- primary MEPM cells using dorsomorphin restored cell proliferation and osteogenic differentiation to wild-type levels. Collectively, these data demonstrate for the first time that Hoxa2 may regulate palate development by inhibiting osteogenic differentiation of palatal mesenchyme via modulating BMP signaling.
Keywords: Hoxa2; RUNX2; bone morphogenetic protein (BMP); cleft palate; osteoblast; osteoprogenitor; proliferation.
Figures


Similar articles
-
Six2 Plays an Intrinsic Role in Regulating Proliferation of Mesenchymal Cells in the Developing Palate.Front Physiol. 2017 Nov 23;8:955. doi: 10.3389/fphys.2017.00955. eCollection 2017. Front Physiol. 2017. PMID: 29218017 Free PMC article.
-
Altered BMP-Smad4 signaling causes complete cleft palate by disturbing osteogenesis in palatal mesenchyme.J Mol Histol. 2021 Feb;52(1):45-61. doi: 10.1007/s10735-020-09922-4. Epub 2020 Nov 7. J Mol Histol. 2021. PMID: 33159638
-
Osteogenic microenvironment affects palatal development through glycolysis.Differentiation. 2023 Sep-Oct;133:1-11. doi: 10.1016/j.diff.2023.05.002. Epub 2023 May 25. Differentiation. 2023. PMID: 37267667
-
Mesenchymal influences on epithelial differentiation in developing systems.J Cell Sci Suppl. 1988;10:195-230. doi: 10.1242/jcs.1988.supplement_10.15. J Cell Sci Suppl. 1988. PMID: 3077937 Review.
-
Bone morphogenetic proteins.Growth Factors. 2004 Dec;22(4):233-41. doi: 10.1080/08977190412331279890. Growth Factors. 2004. PMID: 15621726 Review.
Cited by
-
Identification of sulfur metabolism-related gene signature in osteoarthritis and TM9SF2's sustenance effect on M2 macrophages' phagocytic activity.J Orthop Surg Res. 2024 Jan 13;19(1):62. doi: 10.1186/s13018-023-04384-2. J Orthop Surg Res. 2024. PMID: 38218914 Free PMC article.
-
Isolation and Time-Lapse Imaging of Primary Mouse Embryonic Palatal Mesenchyme Cells to Analyze Collective Movement Attributes.J Vis Exp. 2021 Feb 13;(168):10.3791/62151. doi: 10.3791/62151. J Vis Exp. 2021. PMID: 33645552 Free PMC article.
-
Alx1 Deficient Mice Recapitulate Craniofacial Phenotype and Reveal Developmental Basis of ALX1-Related Frontonasal Dysplasia.Front Cell Dev Biol. 2022 Jan 21;10:777887. doi: 10.3389/fcell.2022.777887. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35127681 Free PMC article.
-
The Forgotten Skeletogenic Condensations: A Comparison of Early Skeletal Development Amongst Vertebrates.J Dev Biol. 2019 Feb 1;7(1):4. doi: 10.3390/jdb7010004. J Dev Biol. 2019. PMID: 30717314 Free PMC article. Review.
-
Transcriptome dynamics of long noncoding RNAs and transcription factors demarcate human neonatal, adult, and human mesenchymal stem cell-derived engineered cartilage.J Tissue Eng Regen Med. 2020 Jan;14(1):29-44. doi: 10.1002/term.2961. Epub 2019 Dec 18. J Tissue Eng Regen Med. 2020. PMID: 31503387 Free PMC article.
References
-
- Barrow J. R., Capecchi M. R. (1999). Compensatory defects associated with mutations in Hoxa1 restore normal palatogenesis to Hoxa2 mutants. Development 126, 5011–5026. Available online at: http://dev.biologists.org/content/126/22/5011.abstract - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

