Decellularization of placentas: establishing a protocol
- PMID: 29185592
- PMCID: PMC5685058
- DOI: 10.1590/1414-431X20176382
Decellularization of placentas: establishing a protocol
Abstract
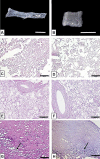
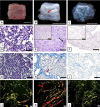
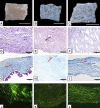
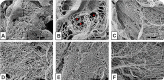
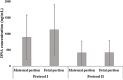
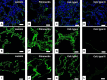
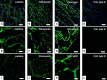
Biological biomaterials for tissue engineering purposes can be produced through tissue and/or organ decellularization. The remaining extracellular matrix (ECM) must be acellular and preserve its proteins and physical features. Placentas are organs of great interest because they are discarded after birth and present large amounts of ECM. Protocols for decellularization are tissue-specific and have not been established for canine placentas yet. This study aimed at analyzing a favorable method for decellularization of maternal and fetal portions of canine placentas. Canine placentas were subjected to ten preliminary tests to analyze the efficacy of parameters such as the type of detergents, freezing temperatures and perfusion. Two protocols were chosen for further analyses using histology, scanning electron microscopy, immunofluorescence and DNA quantification. Sodium dodecyl sulfate (SDS) was the most effective detergent for cell removal. Freezing placentas before decellularization required longer periods of incubation in different detergents. Both perfusion and immersion methods were capable of removing cells. Placentas decellularized using Protocol I (1% SDS, 5 mM EDTA, 50 mM TRIS, and 0.5% antibiotic) preserved the ECM structure better, but Protocol I was less efficient to remove cells and DNA content from the ECM than Protocol II (1% SDS, 5 mM EDTA, 0.05% trypsin, and 0.5% antibiotic).
Figures
Similar articles
-
Fast, robust and effective decellularization of whole human livers using mild detergents and pressure controlled perfusion.Mater Sci Eng C Mater Biol Appl. 2020 Mar;108:110200. doi: 10.1016/j.msec.2019.110200. Epub 2019 Sep 12. Mater Sci Eng C Mater Biol Appl. 2020. PMID: 31923991
-
Tissue engineering of heart valves: biomechanical and morphological properties of decellularized heart valves.J Heart Valve Dis. 2007 Sep;16(5):567-73; discussion 574. J Heart Valve Dis. 2007. PMID: 17944130
-
A novel technique for simultaneous whole-body and multi-organ decellularization: umbilical artery catheterization as a perfusion-based method in a sheep foetus model.Int J Exp Pathol. 2015 Apr;96(2):116-32. doi: 10.1111/iep.12124. Int J Exp Pathol. 2015. PMID: 26031202 Free PMC article.
-
[Research progress of decellularization and application in tissue engineering].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013 Aug;27(8):950-4. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013. PMID: 24171349 Review. Chinese.
-
Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance.Methods. 2015 Aug;84:25-34. doi: 10.1016/j.ymeth.2015.03.005. Epub 2015 Mar 16. Methods. 2015. PMID: 25791470 Review.
Cited by
-
PLLA/GO Scaffolds Filled with Canine Placenta Hydrogel and Mesenchymal Stem Cells for Bone Repair in Goat Mandibles.J Funct Biomater. 2024 Oct 20;15(10):311. doi: 10.3390/jfb15100311. J Funct Biomater. 2024. PMID: 39452609 Free PMC article.
-
Canine Placenta Recellularized Using Yolk Sac Cells with Vascular Endothelial Growth Factor.Biores Open Access. 2018 Jul 1;7(1):101-106. doi: 10.1089/biores.2018.0014. eCollection 2018. Biores Open Access. 2018. PMID: 30065855 Free PMC article.
-
Placental-Derived Biomaterials and Their Application to Wound Healing: A Review.Bioengineering (Basel). 2023 Jul 12;10(7):829. doi: 10.3390/bioengineering10070829. Bioengineering (Basel). 2023. PMID: 37508856 Free PMC article. Review.
-
Human placental extract: the feasibility of translation from basic science into clinical practice.Ann Transl Med. 2020 Mar;8(5):156. doi: 10.21037/atm.2020.01.50. Ann Transl Med. 2020. PMID: 32310247 Free PMC article. No abstract available.
-
Decellularized human amniotic membrane scaffolds: influence on the biological behavior of dental pulp stem cells.BMC Oral Health. 2024 Mar 27;24(1):394. doi: 10.1186/s12903-024-04130-y. BMC Oral Health. 2024. PMID: 38539149 Free PMC article.
References
-
- González-Andrades M, Carriel V, Rivera-Izquierdo M, Garzón I, González-Andrades E, Medialdea S, et al. Effects of Detergent-Based Protocols on Decellularization of Corneas With Sclerocorneal Limbus. Evaluation of Regional Differences. Transl Vis Sci Technol. 2015;4:1–13. doi: 10.1167/tvst.4.2.13. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

